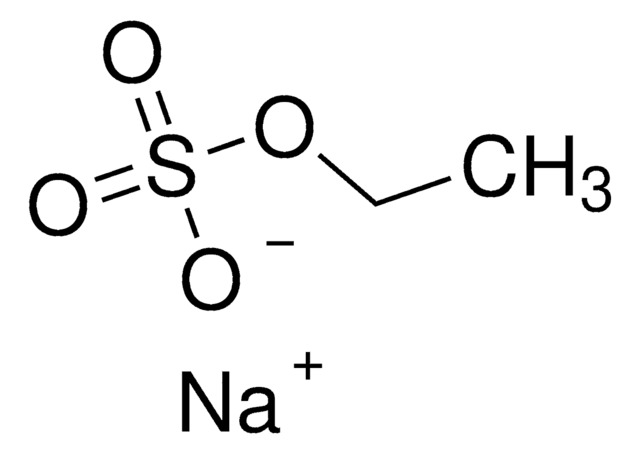
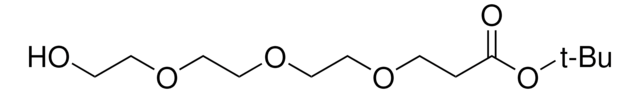
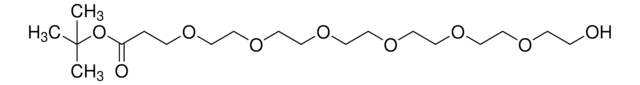
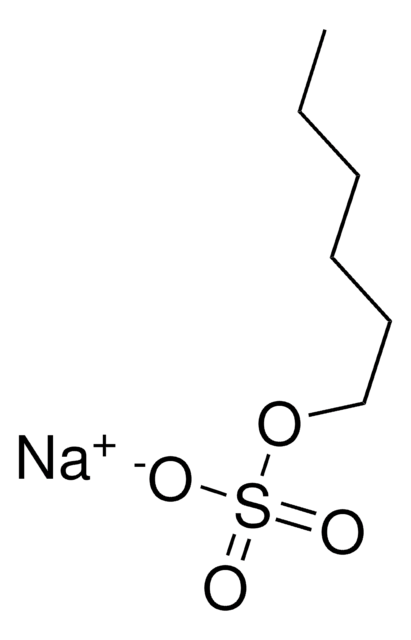
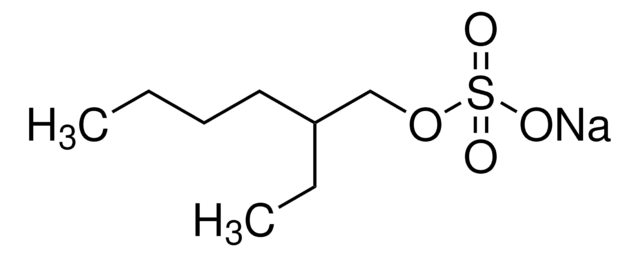
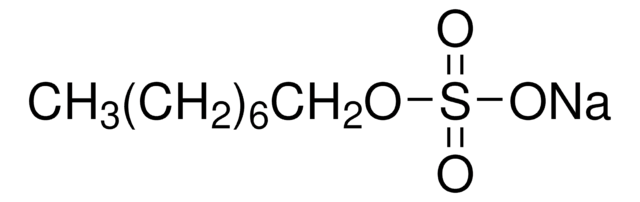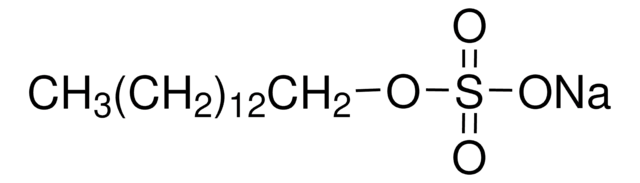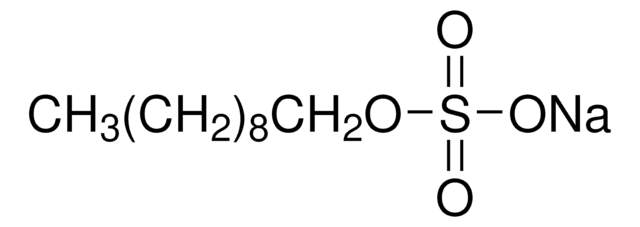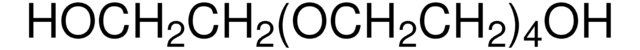推薦產品
化驗
≥95%
形狀
liquid
反應適用性
reagent type: cross-linking reagent
折射率
n/D 1.4492
密度
1.04746 g/mL
官能基
ester
hydroxyl
儲存溫度
2-8°C
SMILES 字串
O=C(OC(C)(C)C)CCOCCOCCOCCOCCO
InChI
1S/C15H30O7/c1-15(2,3)22-14(17)4-6-18-8-10-20-12-13-21-11-9-19-7-5-16/h16H,4-13H2,1-3H3
InChI 密鑰
FJRDXEGYAVAMLB-UHFFFAOYSA-N
應用
This heterobifunctional, PEGylated crosslinker features a hydroxyl group at one end and t-butyl-protected carboxylic acid at the other, which can be deprotected with acidic conditions. The hydrophillic PEG linker facilitates solubility in biological applications. Hydroxy-PEG4-t-butyl ester can be used for bioconjugation or as a building block for synthesis of small molecules, conjugates of small molecules and/or biomolecules, or other tool compounds for chemical biology and medicinal chemistry that require ligation. Examples of applications include its synthetic incorporation into antibody-drug conjugates or proteolysis-targeting chimeras (PROTAC® molecules) for targeted protein degradation.
其他說明
法律資訊
PROTAC is a registered trademark of Arvinas Operations, Inc., and is used under license
相關產品
產品號碼
描述
訂價
儲存類別代碼
10 - Combustible liquids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
Michael L Miller et al.
Journal of medicinal chemistry, 47(20), 4802-4805 (2004-09-17)
To develop effective taxane-antibody immunoconjugates, we have prepared a series of modified taxanes that have both improved toxicity and solubility in aqueous systems as compared to paclitaxel (1a). These taxanes have been modified at either the C-10 or C-7 position
Synthesis of novel heterobifunctional isocyanato cross-linkers and their applications for the preparation of 10-hydroxycamptothecin and SN-38 conjugates with melanotransferrin P97.
Li Z, et al.
Synthetic Communications, 37(11) (2007)
Syntheses and characterizations of novel pyrrolocoumarin probes for SNAP-tag labeling technology.
Mei D, et al
Tetrahedron, 67(12), 2251-2259 (2011)
A new route for the synthesis of 1-amino-3,6,9,12-?tetraoxapentadecan-15-oic acid.
Wu X, et al.
J. Chem. Res. (M), 40(6), 368-370 (2016)
Nico Rublack et al.
Journal of nucleic acids, 2011, 805253-805253 (2011-10-21)
Nowadays, RNA synthesis has become an essential tool not only in the field of molecular biology and medicine, but also in areas like molecular diagnostics and material sciences. Beyond synthetic RNAs for antisense, aptamer, ribozyme, and siRNA technologies, oligoribonucleotides carrying
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務