W381918
L-Arginine
99%, FCC, FG
Synonym(s):
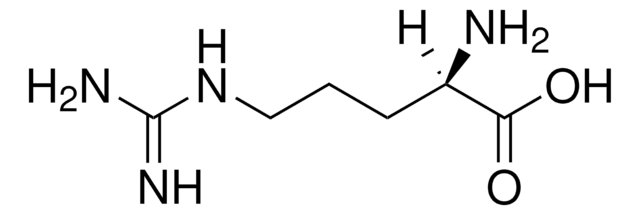
(S)-2-Amino-5-guanidinopentanoic acid
About This Item
Recommended Products
biological source
synthetic
Quality Level
grade
FG
agency
meets purity specifications of JECFA
reg. compliance
EU Regulation 1334/2008 & 872/2012
FCC
FDA 21 CFR 172.320
assay
99%
form
powder or crystals
optical activity
[α]20/D +27°, c = 8 in 6 M HCl
mp
222 °C (dec.) (lit.)
application(s)
flavors and fragrances
documentation
see Safety & Documentation for available documents
food allergen
no known allergens
organoleptic
faint
SMILES string
N[C@@H](CCCNC(N)=N)C(O)=O
InChI
1S/C6H14N4O2/c7-4(5(11)12)2-1-3-10-6(8)9/h4H,1-3,7H2,(H,11,12)(H4,8,9,10)/t4-/m0/s1
InChI key
ODKSFYDXXFIFQN-BYPYZUCNSA-N
Gene Information
human ... NOS1(4842) , NOS2(4843)
rat ... Ppm1a(24666)
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- Metabolomic approaches to dissect dysregulated metabolism in the progression of pre-diabetes to T2DM.: This study employs L-Arginine to examine metabolic changes during the transition from pre-diabetes to type 2 diabetes mellitus, offering new targets for early intervention (Ji et al., 2024 Jun). Link to the article.
- Amino acid profile alteration in age-related atrial fibrillation.: Investigates the role of L-Arginine in cardiovascular aging, highlighting its potential in developing therapeutic strategies against atrial fibrillation associated with aging (Huang et al., 2024 Mar). Link to the article.
Biochem/physiol Actions
Storage Class
11 - Combustible Solids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service