211370
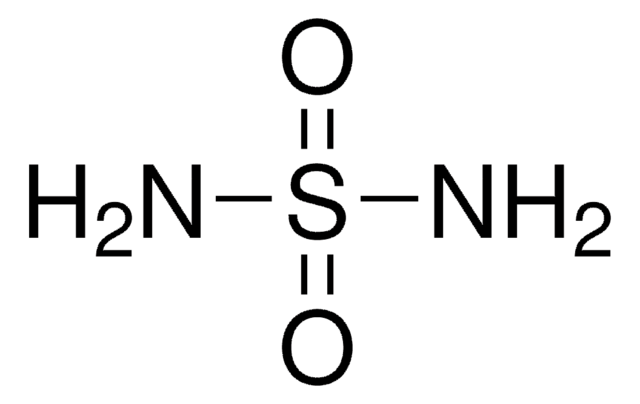
Sulfamide
99%
Synonym(s):
Imidosulfamic acid, Sulfamoylamine, Sulfonyl diamide, Sulfuryl amide, Sulfuryl diamide
About This Item
Recommended Products
Quality Level
assay
99%
mp
90-92 °C (lit.)
solubility
water: soluble 50 mg/mL, clear, colorless to faintly yellow
density
1.611 g/mL at 25 °C (lit.)
SMILES string
NS(N)(=O)=O
InChI
1S/H4N2O2S/c1-5(2,3)4/h(H4,1,2,3,4)
InChI key
NVBFHJWHLNUMCV-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- Schiff bases of the type ArCH=NSO2NH2
- 1H,3H-2,1,3-benzothiadiazin-4-one-2,2-dioxide (BTDD)
- sulfamide analogs of oleoylethanolamide analogs in a study of PPARα activation.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 211370-1G | |
| 211370-5G | 4061838771773 |
| 211370-25G | 4061832418872 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service