Key Documents
D3876
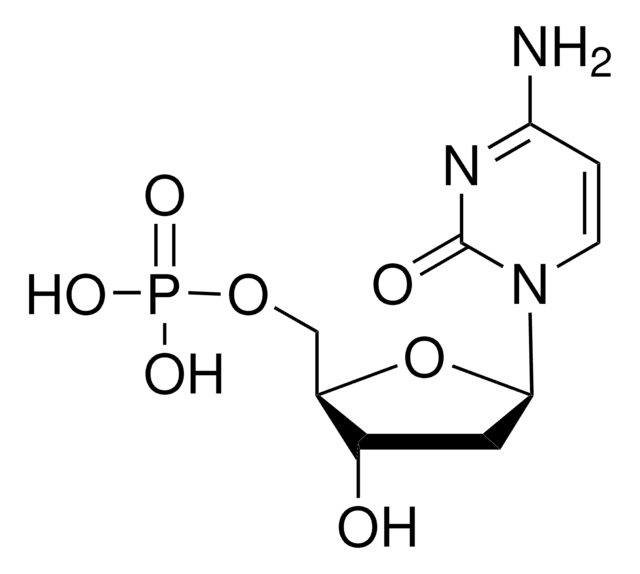
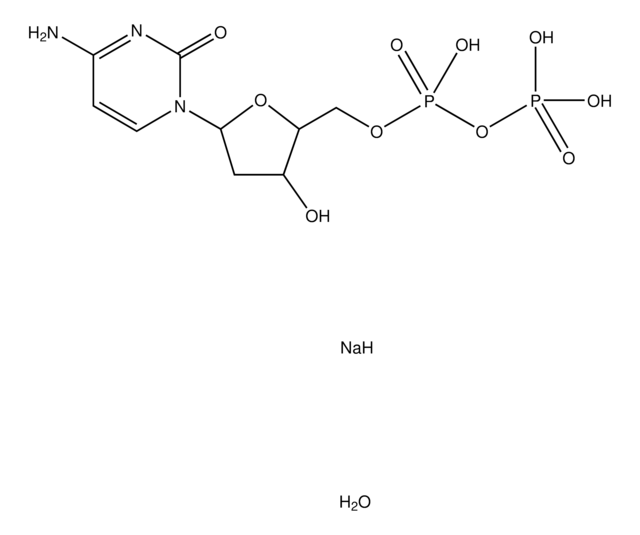
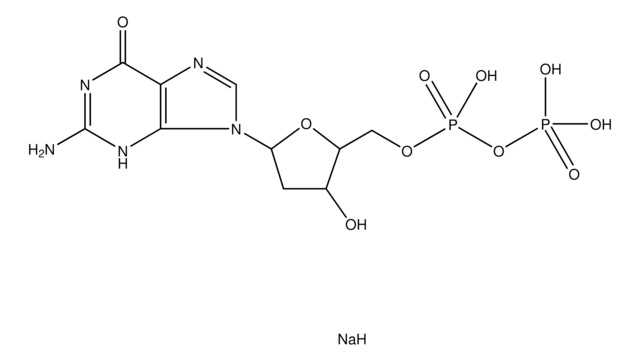
2′-Deoxyuridine 5′-monophosphate disodium salt
Sigma Grade
Synonim(y):
dUMP
About This Item
Polecane produkty
pochodzenie biologiczne
synthetic (organic)
Poziom jakości
klasa czystości
Sigma Grade
Próba
≥98% (HPLC)
Postać
powder
temp. przechowywania
−20°C
ciąg SMILES
[Na].OC1CC(OC1COP(O)(O)=O)N2C=CC(=O)NC2=O
InChI
1S/C9H13N2O8P.Na.H/c12-5-3-8(11-2-1-7(13)10-9(11)14)19-6(5)4-18-20(15,16)17;;/h1-2,5-6,8,12H,3-4H2,(H,10,13,14)(H2,15,16,17);;
Klucz InChI
WXIVKKBDJOCRNB-UHFFFAOYSA-N
Opis ogólny
Zastosowanie
- in ultraperformance liquid chromatography-tandem mass spectrometry (UPLC/MS/MS) assay
- to stimulate proliferation of peripheral blood mononuclear cell (PBMCs) exposed to influenza virus antigen
- in thymidylate synthase activity assay Helicobacter pylori
Działania biochem./fizjol.
Hasło ostrzegawcze
Warning
Zwroty wskazujące rodzaj zagrożenia
Zwroty wskazujące środki ostrożności
Klasyfikacja zagrożeń
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organy docelowe
Respiratory system
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Środki ochrony indywidualnej
dust mask type N95 (US), Eyeshields, Gloves
Certyfikaty analizy (CoA)
Poszukaj Certyfikaty analizy (CoA), wpisując numer partii/serii produktów. Numery serii i partii można znaleźć na etykiecie produktu po słowach „seria” lub „partia”.
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej