Y0000001
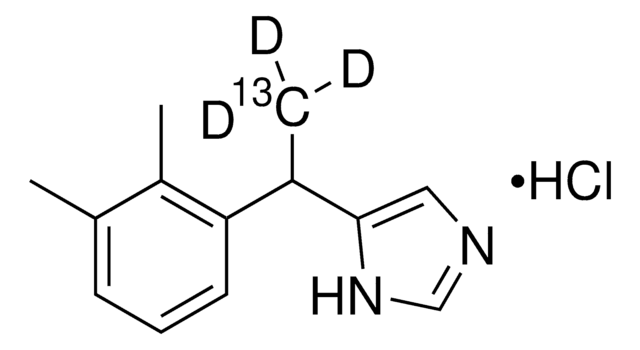
Detomidine hydrochloride
European Pharmacopoeia (EP) Reference Standard
About This Item
Polecane produkty
klasa czystości
pharmaceutical primary standard
rodzina API
dexmedetomidine, medetomidine
producent / nazwa handlowa
EDQM
Zastosowanie
pharmaceutical (small molecule)
Format
neat
temp. przechowywania
2-8°C
ciąg SMILES
[Cl-].[nH]1cncc1Cc2c(c(ccc2)C)C.[H+]
InChI
1S/C12H14N2.ClH/c1-9-4-3-5-11(10(9)2)6-12-7-13-8-14-12;/h3-5,7-8H,6H2,1-2H3,(H,13,14);1H
Klucz InChI
OIWRDXKNDCJZSM-UHFFFAOYSA-N
Opis ogólny
Zastosowanie
Opakowanie
Inne uwagi
produkt powiązany
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Wybierz jedną z najnowszych wersji:
Certyfikaty analizy (CoA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Dokumenty section.
Proszę o kontakt, jeśli potrzebna jest pomoc Obsługa Klienta
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej