Kluczowe dokumenty
T3383
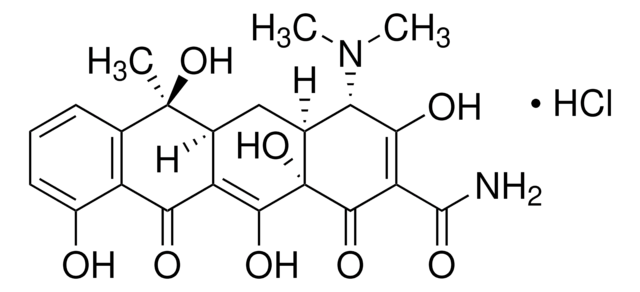
Tetracycline hydrochloride
≥95% (European Pharmacopoeia HPLC assay)
Synonim(y):
4-Epitetracycline hydrochloride, Achromycin hydrochloride
About This Item
Polecane produkty
Poziom jakości
Próba
≥95% (European Pharmacopoeia HPLC assay)
Formularz
powder
warunki przechowywania
(Keep container tightly closed in a dry and well-ventilated place.)
kolor
yellow
mp
220-223 °C (lit.)
spektrum działania antybiotyku
Gram-negative bacteria
Gram-positive bacteria
Tryb działania
protein synthesis | interferes
temp. przechowywania
−20°C
ciąg SMILES
Cl.CN(C)[C@H]1[C@@H]2C[C@H]3C(=C(O)[C@]2(O)C(=O)C(C(N)=O)=C1O)C(=O)c4c(O)cccc4[C@@]3(C)O
InChI
1S/C22H24N2O8.ClH/c1-21(31)8-5-4-6-11(25)12(8)16(26)13-9(21)7-10-15(24(2)3)17(27)14(20(23)30)19(29)22(10,32)18(13)28;/h4-6,9-10,15,25,27-28,31-32H,7H2,1-3H3,(H2,23,30);1H/t9-,10-,15-,21+,22-;/m0./s1
Klucz InChI
XMEVHPAGJVLHIG-FMZCEJRJSA-N
Szukasz podobnych produktów? Odwiedź Przewodnik dotyczący porównywania produktów
Powiązane kategorie
Opis ogólny
Zastosowanie
Działania biochem./fizjol.
Mode of Resistance: The effects are inactivated via a loss of cell wall permeability.
Antimicrobial spectrum: Includes a wide range of antimicrobial activity against gram-positive and gram-negative bacteria.
Przestroga
Uwaga dotycząca przygotowania
Inne uwagi
Hasło ostrzegawcze
Warning
Zwroty wskazujące rodzaj zagrożenia
Zwroty wskazujące środki ostrożności
Klasyfikacja zagrożeń
Aquatic Acute 1 - Aquatic Chronic 2 - Eye Irrit. 2 - Repr. 2 - Skin Irrit. 2 - STOT SE 3
Organy docelowe
Respiratory system
Kod klasy składowania
11 - Combustible Solids
Klasa zagrożenia wodnego (WGK)
WGK 2
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Środki ochrony indywidualnej
dust mask type N95 (US), Eyeshields, Gloves
Wybierz jedną z najnowszych wersji:
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej