1278007
USP
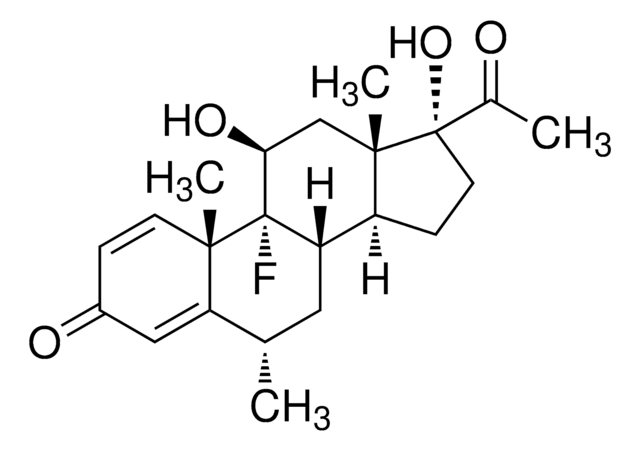
Fluorometholone
United States Pharmacopeia (USP) Reference Standard
동의어(들):
11β,17α-Dihydroxy-9-fluoro-6-methyl-1,4-pregnadiene-3,20-dione
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C22H29FO4
CAS Number:
Molecular Weight:
376.46
MDL number:
UNSPSC 코드:
41116107
PubChem Substance ID:
NACRES:
NA.24
추천 제품
Grade
pharmaceutical primary standard
API family
fluorometholone
제조업체/상표
USP
응용 분야
pharmaceutical (small molecule)
형식
neat
SMILES string
C[C@H]1C[C@H]2[C@@H]3CC[C@](O)(C(C)=O)[C@@]3(C)C[C@H](O)[C@]2(F)[C@@]4(C)C=CC(=O)C=C14
InChI
1S/C22H29FO4/c1-12-9-17-15-6-8-21(27,13(2)24)20(15,4)11-18(26)22(17,23)19(3)7-5-14(25)10-16(12)19/h5,7,10,12,15,17-18,26-27H,6,8-9,11H2,1-4H3/t12-,15-,17-,18-,19-,20-,21-,22-/m0/s1
InChI key
FAOZLTXFLGPHNG-KNAQIMQKSA-N
유전자 정보
human ... NR3C1(2908)
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.For further information and support please go to the website of the issuing Pharmacopoeia.
애플리케이션
Fluorometholone USP reference standard, intended for use in specified quality tests and assays as specified in the USP compendia. Also, for use with USP monographs such as:
- Fluorometholone Acetate
- Fluorometholone Ophthalmic Suspension
- Hydrocortisone Hemisuccinate
- Hydrocortisone Sodium Succinate for Injection
- Methylprednisolone Hemisuccinate
- Methylprednisolone Sodium Succinate for Injection
생화학적/생리학적 작용
Clinically significant in allergic conjunctivitis and as anti-inflammatory following cataract surgery.
분석 메모
These products are for test and assay use only. They are not meant for administration to humans or animals and cannot be used to diagnose, treat, or cure diseases of any kind.
기타 정보
Sales restrictions may apply.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
Asimina Mataftsi et al.
The British journal of ophthalmology, 95(11), 1531-1533 (2011-02-08)
Although fluorometholone (FML) is considered a steroid of minimal ocular penetration, reports in children have shown dose-dependent intraocular pressure (IOP) rise. The authors aimed to assess whether reducing regimens of FML for paediatric ocular surface disease have sustained clinically significant
Tear menisci after laser in situ keratomileusis with mechanical microkeratome and femtosecond laser.
Wenjia Xie et al.
Investigative ophthalmology & visual science, 55(9), 5806-5812 (2014-08-26)
To investigate the effect on tear menisci after laser in situ keratomileusis (LASIK) with flap creation by either microkeratome or femtosecond laser. Sixty eyes of 30 myopes were analyzed. Fifteen patients underwent LASIK with Moria II microkeratome, and the other
Hua Gao et al.
Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie, 252(6), 963-968 (2014-05-07)
To evaluate the feasibility of partial lamellar keratoplasty (LK) for treatment of peripheral corneal disease (PCD) using a graft from the corneoscleral rim preserved in glycerin. Patients who underwent LK for PCD at Shandong Eye Hospital from January 2006 to
Miaomiao Zhang et al.
Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie, 252(6), 983-987 (2014-03-29)
To analyze the efficacy of selective laser trabeculoplasty (SLT) on silicone oil-induced secondary glaucoma in terms of intraocular pressure (IOP). 42 patients (42 eyes) with silicone oil-induced secondary glaucoma were selected, and SLT was performed with 360° of the trabecular
Lingmin He et al.
JAMA ophthalmology, 133(1), 51-59 (2014-10-17)
Wavefront-guided (WFG) and wavefront-optimized (WFO) platforms for refractive surgery are designed for improved visual outcomes. It is unclear which treatment profile is superior for patients undergoing photorefractive keratectomy (PRK). To compare the safety, efficacy, predictability, stability, and higher-order aberrations in
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.