1005004
USP
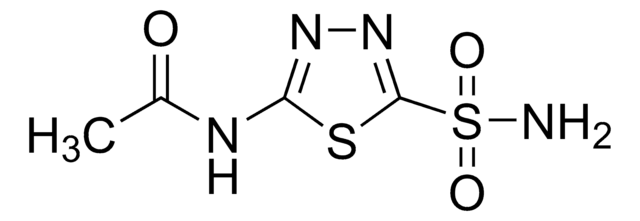
Acetazolamide
United States Pharmacopeia (USP) Reference Standard
동의어(들):
5-Acetamido-1,3,4-thiadiazole-2-sulfonamide, N-(5-Sulfamoyl-1,3,4-thiadiazol-2-yl)acetamide, N-(5-[Aminosulfonyl]-1,3,4-thiadiazol-2-yl)acetamide
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C4H6N4O3S2
CAS Number:
Molecular Weight:
222.25
MDL number:
UNSPSC 코드:
41116107
PubChem Substance ID:
NACRES:
NA.24
추천 제품
Grade
pharmaceutical primary standard
API family
acetazolamide
제조업체/상표
USP
응용 분야
pharmaceutical (small molecule)
형식
neat
SMILES string
CC(=O)Nc1nnc(s1)S(N)(=O)=O
InChI
1S/C4H6N4O3S2/c1-2(9)6-3-7-8-4(12-3)13(5,10)11/h1H3,(H2,5,10,11)(H,6,7,9)
InChI key
BZKPWHYZMXOIDC-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia.For further information and support please go to the website of the issuing Pharmacopoeia.
애플리케이션
Acetazolamide USP reference standard, intended for use in specified quality tests and assays as specified in the USP compendia. Also, for use with USP monographs such as:
- Acetazolamide Compounded Oral Suspension
- Acetazolamide Extended-Release Capsules
- Acetazolamide for Injection
- Acetazolamide Tablets
Carbonic anhydrase inhibitor; increases cerebral blood flow.
생화학적/생리학적 작용
Inhibits water permeability of membranes by interacting with aquaporins
분석 메모
These products are for test and assay use only. They are not meant for administration to humans or animals and cannot be used to diagnose, treat, or cure diseases of any kind.
기타 정보
Sales restrictions may apply.
관련 제품
제품 번호
설명
가격
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
Gladys E Granero et al.
Expert opinion on drug delivery, 7(8), 943-953 (2010-06-23)
Acetazolamide (ACZ), a carbonic anhydrase inhibitor (CAI), and other oral CAIs have been an integral part of antiglaucoma therapy for > 40 years. ACZ is used orally for the reduction of intraocular pressure in patients suffering from glaucoma. However, this
David E Leaf et al.
Journal of applied physiology (Bethesda, Md. : 1985), 102(4), 1313-1322 (2006-10-07)
Acetazolamide, a potent carbonic anhydrase (CA) inhibitor, is the most commonly used and best-studied agent for the amelioration of acute mountain sickness (AMS). The actual mechanisms by which acetazolamide reduces symptoms of AMS, however, remain unclear. Traditionally, acetazolamide's efficacy has
Indu Pal Kaur et al.
International journal of pharmaceutics, 248(1-2), 1-14 (2002-11-14)
Through this review it is contemplated that acetazolamide (ACZ), an age-old treatment for glaucoma with a myriad of side effects and inadequate topical effectiveness, may be formulated into a topically effective agent by utilizing various newer formulation approaches of ocular
G Settakis et al.
European journal of neurology, 10(6), 609-620 (2003-12-04)
Pathologic processes affecting the brain vessels may damage cerebral vasodilatory capacity. Early detection of cerebral dysfunction plays an important role in the prevention of cerebrovascular diseases. In recent decades acetazolamide (AZ) has frequently been used for this purpose. In the
Neil D Ritchie et al.
Journal of travel medicine, 19(5), 298-307 (2012-09-05)
Acetazolamide has been reported to be effective in the prevention of acute mountain sickness (AMS). Our aim was to conduct a systematic review of randomized, placebo-controlled trials of acetazolamide in the prevention of AMS. Studies were identified by searching the
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.