PHR1577
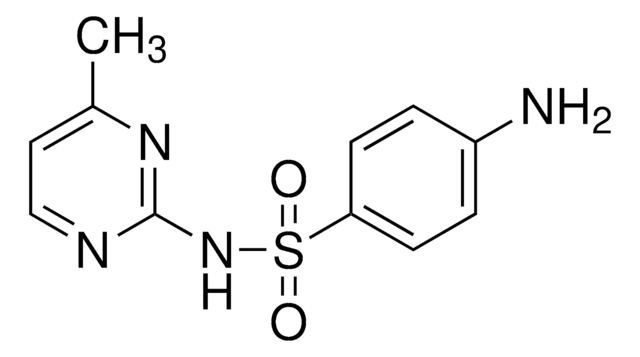
Sulfamerazine
Pharmaceutical Secondary Standard; Certified Reference Material
동의어(들):
Sulfamerazine, 4-Amino-N-(4-methyl-2-pyrimidinyl)benzenesulfonamide, N1-(4-Methylpyrimidin-2-yl)sulfanilamide
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
실험식(Hill 표기법):
C11H12N4O2S
CAS Number:
Molecular Weight:
264.30
Beilstein:
249133
EC Number:
MDL number:
UNSPSC 코드:
41116107
PubChem Substance ID:
NACRES:
NA.24
추천 제품
Grade
certified reference material
pharmaceutical secondary standard
Quality Level
Agency
traceable to Ph. Eur. S2000000
traceable to USP 1628007
API family
sulfamerazine
CofA
current certificate can be downloaded
포장
pkg of 1 g
기술
HPLC: suitable
gas chromatography (GC): suitable
응용 분야
pharmaceutical (small molecule)
형식
neat
저장 온도
2-30°C
SMILES string
Cc1ccnc(NS(=O)(=O)c2ccc(N)cc2)n1
InChI
1S/C11H12N4O2S/c1-8-6-7-13-11(14-8)15-18(16,17)10-4-2-9(12)3-5-10/h2-7H,12H2,1H3,(H,13,14,15)
InChI key
QPPBRPIAZZHUNT-UHFFFAOYSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards. Sulfamerazine belongs to the class of sulfonamides, used as an antibacterial agent to treat infections, especially for patients intolerant to antibiotics.
애플리케이션
Sulfamerazine may be used as a pharmaceutical reference standard for the quantification of the analyte in pharmaceutical formulations using spectrophototmetric technique.
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
분석 메모
These secondary standards offer multi-traceability to the USP, EP and BP primary standards, where they are available.
기타 정보
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
각주
To see an example of a Certificate of Analysis for this material enter LRAA4493 in the Documents slot below. This is an example certificate only and may not be the lot that you receive.
추천 제품
Find a digital Reference Material for this product available on our online platform ChemisTwin® for NMR. You can use this digital equivalent on ChemisTwin® for your sample identity confirmation and compound quantification (with digital external standard). An NMR spectrum of this substance can be viewed and an online comparison against your sample can be performed with a few mouseclicks. Learn more here and start your free trial.
신호어
Warning
유해 및 위험 성명서
예방조치 성명서
Hazard Classifications
Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
A sensitive spectrophotometric method for the determination of sulfonamides in pharmaceutical preparations
Nagaraja P, et al.
Acta pharmaceutica, 57(3), 333-342 (2007)
Marco De Liguoro et al.
Ecotoxicology and environmental safety, 164, 84-91 (2018-08-12)
In livestock farming, sulfonamides (SAs) are used prophylactically and simultaneously in large numbers of animals. Therefore, traces of these compounds, alone or in combination, have been repeatedly detected in the environment. Synergistic interactions among chemicals in such mixtures represent an
Padmarjaiah Nagaraja et al.
European journal of pharmaceutics and biopharmaceutics : official journal of Arbeitsgemeinschaft fur Pharmazeutische Verfahrenstechnik e.V, 53(2), 187-192 (2002-03-07)
A rapid, selective and simple spectrophotometric method for the determination of sulfa-drugs is described. The method is based on the formation of violet colored azo product by the diazotization of sulfonamides, viz. sulfathiazole (SFT), sulfadiazine (SFD), sulfacetamide (SFA), sulfamethoxazole (SFMx)
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.