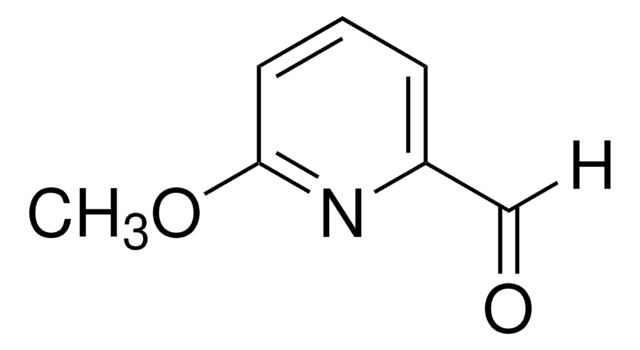
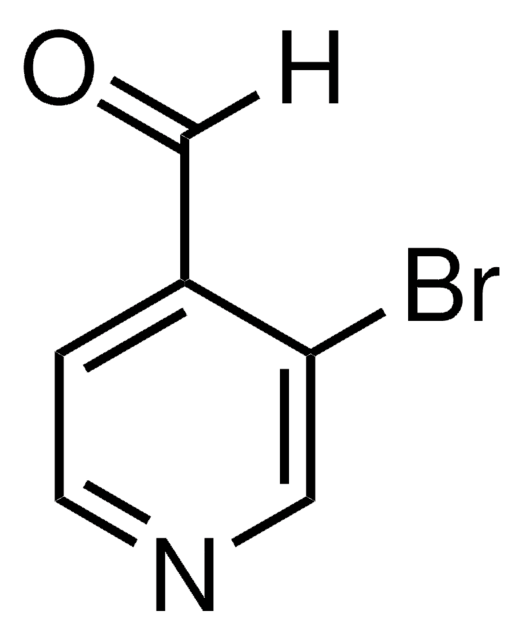
525693
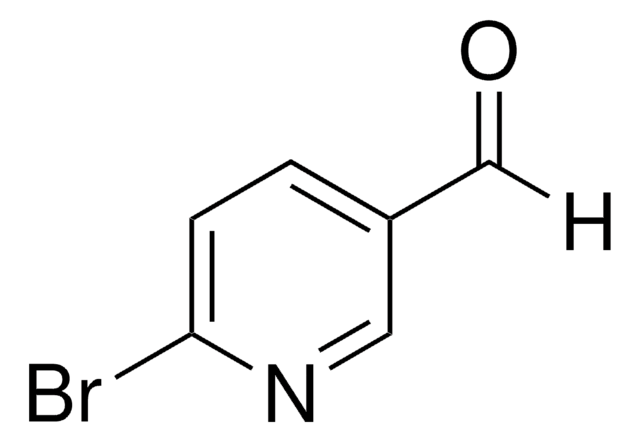
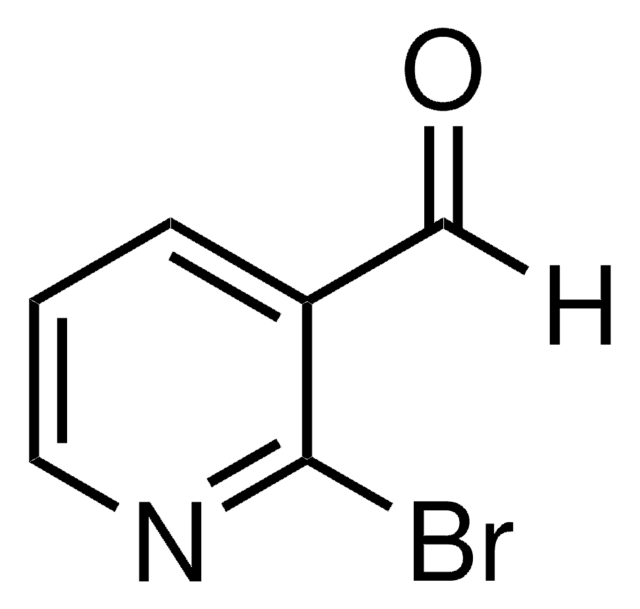
6-Bromo-2-pyridinecarboxaldehyde
97%
동의어(들):
2-Bromo-6-formylpyridine, 2-Bromopyridine-6-carboxaldehyde, 2-Bromopyridyl-6-carboxaldehyde, 2-Formyl-6-bromopyridine, 6-Bromo-2-formylpyridine, 6-Bromo-2-pyridinecarbaldehyde, 6-Bromopicolinaldehyde, 6-Bromopyridine-2-aldehyde
로그인조직 및 계약 가격 보기
모든 사진(2)
About This Item
실험식(Hill 표기법):
C6H4BrNO
CAS Number:
Molecular Weight:
186.01
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Quality Level
분석
97%
mp
81-85 °C (lit.)
작용기
aldehyde
bromo
저장 온도
2-8°C
SMILES string
[H]C(=O)c1cccc(Br)n1
InChI
1S/C6H4BrNO/c7-6-3-1-2-5(4-9)8-6/h1-4H
InChI key
QWFHFNGMCPMOCD-UHFFFAOYSA-N
일반 설명
6-Bromo-2-pyridinecarboxaldehyde is a pyridine derivative. It participates in the synthesis of meso-substituted trans-A2B2-porphyrin.
애플리케이션
6-Bromo-2-pyridinecarboxaldehyde may be used in the synthesis of:
- 6-([2,2′-bi(1,3-dithiolylidene)]-4-yl)picolinaldehyde
- tris(2-pyridylmethyl)amine (Tpy) derivative having Tpy core bearing a 3-formyl-phenyl substituent on one of the three arms
- syn- and anti- forms of 4-(6-bromopyridin-2-yl)-3-methyl-3-phenyloxetan-2-one
- 6-(2,4,6-triisopropylphenyl)-2-pyridinecarboxaldehyde
Substrate used in a study of a rhodium-catalyzed, reductive aldol coupling with divinyl ketones leading to syn ß-hydroxyenones.
Useful building block for Tris[(pyridyl)methyl]amine ligands.
신호어
Warning
유해 및 위험 성명서
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
표적 기관
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
개인 보호 장비
dust mask type N95 (US), Eyeshields, Gloves
이미 열람한 고객
Steric and Electronic Effects in the Formation and Carbon Disulfide Reactivity of Dinuclear Nickel Complexes Supported by Bis (iminopyridine) Ligands.
Bheemaraju A, et al.
Organometallics, 32(10), 2952-2962 (2013)
Francesca A Scaramuzzo et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 19(49), 16809-16813 (2013-11-01)
Reliable determination of the enantiomeric excess of free amino acids can be obtained by measuring the induced circular dichroism of a multicomponent assembly formed by a modified tris(2-pyridylmethyl)amine ligand, a zinc salt, and the amino acid of interest. The systems
Awatef Ayadi et al.
Beilstein journal of organic chemistry, 11, 1379-1391 (2015-10-02)
The synthesis and full characterization of two tetrathiafulvalene-appended azine ligands, namely 2-([2,2'-bi(1,3-dithiolylidene)]-4-yl)-6-((2,4-dinitrophenyl)hydrazono)methyl)pyridine (L1) and 5-([2,2'-bi(1,3-dithiolylidene)]-4-yl)-2-((2,4-dinitrophenyl)hydrazono)methyl)pyridine (L2) are described. The crystal structure of ligand L1 indicates that the ligand is completely planar with the presence of a strong intramolecular N3-H3···O1 hydrogen
Synthetic methodologies leading to porphyrin-quinone conjugates.
Cardoso MFDC, et al.
Journal of Porphyrins and Phthalocyanines, 1-23 (2015)
Soo Bong Han et al.
Organic letters, 8(24), 5657-5660 (2006-11-17)
Catalytic hydrogenation of divinyl ketones 1a and 1e in the presence of diverse aldehydes 2a-e at ambient temperature and pressure using cationic rhodium catalysts ligated by tri-2-furyl phosphine enables formation of aldol products 3a-e and 5a-e, respectively, with high levels
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.