모든 사진(1)
About This Item
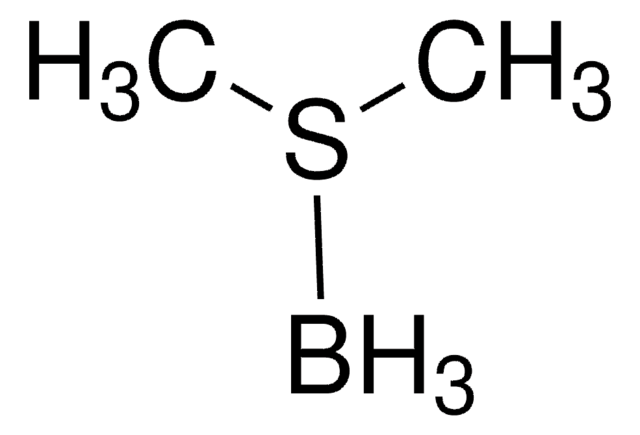
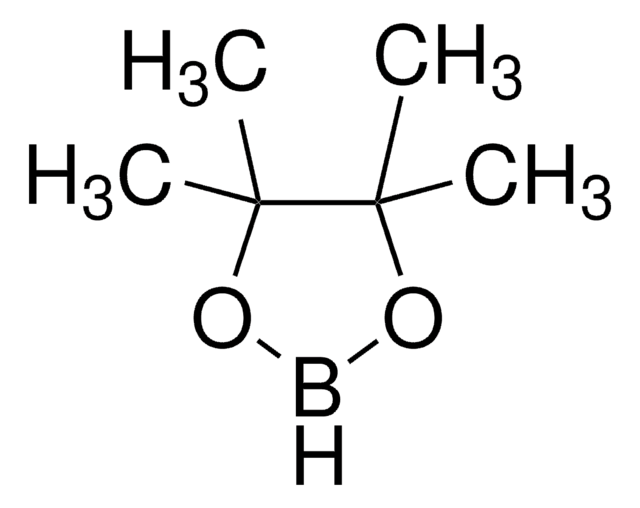
실험식(Hill 표기법):
C8H15B
CAS Number:
Molecular Weight:
122.02
Beilstein:
605509
MDL number:
UNSPSC 코드:
12352005
PubChem Substance ID:
NACRES:
NA.22
추천 제품
반응 적합성
reagent type: reductant
농도
0.4 M in hexanes
bp
68-70 °C
density
0.691 g/mL at 25 °C
SMILES string
B1C2CCCC1CCC2
InChI
1S/C8H15B/c1-3-7-5-2-6-8(4-1)9-7/h7-9H,1-6H2/t7-,8+
InChI key
FEJUGLKDZJDVFY-OCAPTIKFSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
애플리케이션
Protecting group for alkenes†
Reactant for:
Reactant for:
- Linear SPPS synthesis of ubiquitin derivatives
- Copper-catalyzed cross-coupling reactions of organoboron compounds with primary alkyl halides and pseudohalides
- Intramolecular insertion of alkenes into palladium-nitrogen bonds
- Preparation of (phosphonoacetyl)ornithine to study effect on arginine biosynthetic genes in yeast
- Hetero-Diels-Alder reaction for synthesis of spirocyclic alkaloids
신호어
Danger
유해 및 위험 성명서
Hazard Classifications
Aquatic Chronic 2 - Asp. Tox. 1 - Eye Irrit. 2 - Repr. 2 - Skin Irrit. 2 - STOT RE 1 Inhalation - STOT SE 3 - Water-react 1
표적 기관
Central nervous system, Nervous system
Storage Class Code
4.3 - Hazardous materials which set free flammable gases upon contact with water
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
이미 열람한 고객
Yi Luan et al.
Organic letters, 13(9), 2510-2513 (2011-04-09)
Diazo esters, arylboranes, and carbamoyl imines undergo a 3-component Mannich condensation reaction. Catalyzed by Cu(II) salts, the reaction affords the corresponding isocyanate Mannich product: one that can be easily trapped in situ by other nucleophiles. The Mannich condensation corresponds to
A Ganesan
Mini reviews in medicinal chemistry, 6(1), 3-10 (2006-02-07)
Solid-phase synthesis is a powerful tool for achieving high-throughput chemistry. This review discusses recent diverse examples from my group: the solid-phase synthesis of unsymmetrical guanidines, polymer-supported versions of cyclooctadiene and 9-BBN, a triflate-like linker, the synthesis of tetrahydro-beta-carbolines by the
Jefferson D Revell et al.
Organic letters, 7(5), 831-833 (2005-02-25)
1,5-Cyclooctadiene was deprotonated under LICKOR conditions and reacted with Merrifield resin to afford an immobilized cyclooctadiene in high yield. This polymer is effective as a halogen scavenger, while hydroboration leads to a supported 9-BBN analogue. The latter exhibits similar regioselectivity
Rajesh Sardar et al.
Journal of the American Chemical Society, 133(21), 8179-8190 (2011-05-10)
We report a spectroscopic and microscopic investigation of the synthesis of gold nanoparticles (AuNPs) with average sizes of less than 5 nm. The slow reduction and AuNP formation processes that occur by using 9-borabicyclo[3.3.1]nonane (9-BBN) as a reducing agent enabled
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.![9-Borabicyclo[3.3.1]nonane solution 0.5 M in THF](/deepweb/assets/sigmaaldrich/product/structures/180/891/8b64e597-269d-4780-98b6-40889dfd06b9/640/8b64e597-269d-4780-98b6-40889dfd06b9.png)
![9-Borabicyclo[3.3.1]nonane dimer](/deepweb/assets/sigmaaldrich/product/structures/203/431/624973a6-aec1-4b23-b6c4-013285ac418c/640/624973a6-aec1-4b23-b6c4-013285ac418c.png)