おすすめの製品
製品名
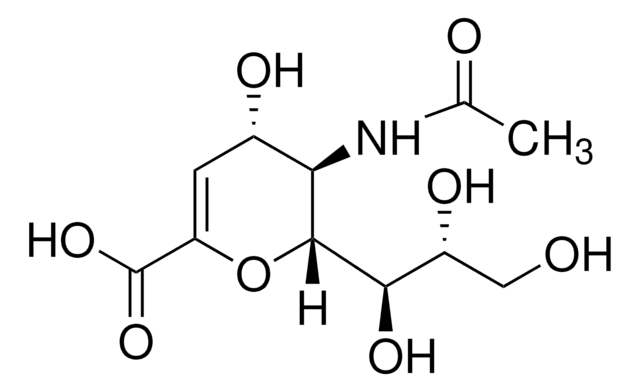
シアスタチンB, lyophilized powder, from microbial
由来生物
microbial
品質水準
フォーム
lyophilized powder
包装
vial of 23.0 μmol
溶解性
H2O: 2.3 mL/vial (for a 10 mM solution)
保管温度
−20°C
SMILES記法
CC(=O)N[C@H]1NC[C@@H]([C@H](O)[C@@H]1O)C(O)=O
InChI
1S/C8H14N2O5/c1-3(11)10-7-6(13)5(12)4(2-9-7)8(14)15/h4-7,9,12-13H,2H2,1H3,(H,10,11)(H,14,15)/t4-,5-,6-,7+/m0/s1
InChI Key
DQTKLICLJUKNCG-ZTYPAOSTSA-N
アプリケーション
適用範囲が広い、シアリダ-ゼ阻害剤
生物化学的/生理学的作用
広域スペクトルのノイラミニダーゼ(シアリダーゼ)インヒビターである、ストレプトマイセス代謝物。
シグナルワード
Warning
危険有害性情報
危険有害性の分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
ターゲットの組織
Respiratory system
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
個人用保護具 (PPE)
dust mask type N95 (US), Eyeshields, Gloves
適用法令
試験研究用途を考慮した関連法令を主に挙げております。化学物質以外については、一部の情報のみ提供しています。 製品を安全かつ合法的に使用することは、使用者の義務です。最新情報により修正される場合があります。WEBの反映には時間を要することがあるため、適宜SDSをご参照ください。
Jan Code
S8063-5MG-PW:
S8063-BULK:
S8063-VAR:
S8063-5MG:
T Takatsu et al.
The Journal of antibiotics, 49(1), 54-60 (1996-01-01)
Novel heparanse inhibitors, A72363 A-1, A-2, and C, were isolated from the culture filtrate of Streptomyces nobilis SANK 60192 by column chromatography on various resinous adsorbents, followed by preparative anion exchange HPLC. Spectroscopic studies revealed that they are diastereomers of
K I Kondo et al.
Natural product letters, 15(6), 371-375 (2002-02-13)
N-Alkyl-3-decarboxy-3-hydroxymethylsiastatin B, N-alkyl analogues of gem-diamine 1-N-iminosugars, is a new family of glycosidase inhibitors that have been synthesized from siastatin B isolated from Streptomyces culture. These compounds were evaluated as glycosidase inhibitors.
Y Nishimura et al.
Bioorganic & medicinal chemistry, 4(1), 91-96 (1996-01-01)
N-Acetylgalactosamine-based 1-N-iminosugars, new types of glycosidase inhibitor have been synthesized by modeling on siastatin B, isolated from a Streptomyces culture. The analogues of siastatin B were proved to be potent inhibitors for alpha-N-acetylgalactosaminidase and/or beta-N-acetylglucosaminidase.
Synthesis and antimetastatic activity of 6-trichloroacetamido and 6-guanidino analogues of siastatin B.
T Satoh et al.
The Journal of antibiotics, 49(3), 321-325 (1996-03-01)
T Kudo et al.
The Journal of antibiotics, 45(6), 954-962 (1992-06-01)
Totally synthetic analogues of siastatin B, optically active 2-acetamido-3,4,5-trihydroxypiperidines having the nitromethyl, aminomethyl and carboxyl branched groups at C-5 have been obtained from D-ribono-1,4-lactone by a stereospecific convergent method. Some analogues showed inhibitory activity against some glycosidases.
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)