おすすめの製品
グレード
pharmaceutical primary standard
APIファミリー
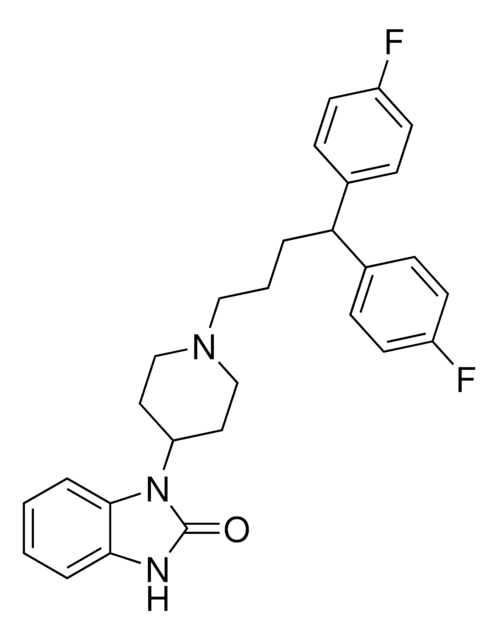
pimozide
メーカー/製品名
EDQM
アプリケーション
pharmaceutical (small molecule)
フォーマット
neat
保管温度
2-8°C
SMILES記法
Fc1ccc(cc1)C(CCCN2CCC(CC2)N3C(=O)Nc4ccccc34)c5ccc(F)cc5
InChI
1S/C28H29F2N3O/c29-22-11-7-20(8-12-22)25(21-9-13-23(30)14-10-21)4-3-17-32-18-15-24(16-19-32)33-27-6-2-1-5-26(27)31-28(33)34/h1-2,5-14,24-25H,3-4,15-19H2,(H,31,34)
InChI Key
YVUQSNJEYSNKRX-UHFFFAOYSA-N
遺伝子情報
human ... DRD1(1812) , DRD2(1813) , DRD3(1814) , DRD4(1815) , DRD5(1816) , HTR2A(3356)
類似した製品をお探しですか? 訪問 製品比較ガイド
関連するカテゴリー
詳細
この製品は薬局方標準品です。発行元の薬局方により製造・供給されています。MSDSを含む製品情報などの詳しい情報は、発行元の薬局方のウェブサイトよりご確認ください。
アプリケーション
Pimozide EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
包装
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
その他情報
Sales restrictions may apply.
シグナルワード
Warning
危険有害性情報
危険有害性の分類
Acute Tox. 4 Oral
保管分類コード
11 - Combustible Solids
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
最新バージョンのいずれかを選択してください:
Cynthia R Lorenzo et al.
American journal of clinical dermatology, 5(5), 339-349 (2004-11-24)
Pimozide is an antipsychotic drug of the diphenylbutylpiperidine class. In the US, it is FDA-approved only as a backup treatment for Gilles de la Tourette syndrome, although it has been used in other countries for many years as a treatment
Tamara Pringsheim et al.
The Cochrane database of systematic reviews, (2)(2), CD006996-CD006996 (2009-04-17)
Neuroleptic drugs with potent D-2 receptor blocking properties have been the traditional treatment for tics caused by Tourette Syndrome. Pimozide is the most studied of these. Use of these medications is declining because of concerns about side effects, and new
Meghana Mothi et al.
The Cochrane database of systematic reviews, 11(11), CD001949-CD001949 (2013-11-07)
Pimozide, formulated in the 1960s, continues to be marketed for the care of people with schizophrenia or related psychoses such as delusional disorder. It has been associated with cardiotoxicity and sudden unexplained death. Electrocardiogram monitoring is now required before and
C L Colvin et al.
Drug intelligence & clinical pharmacy, 19(6), 421-424 (1985-06-01)
The orphan drug pimozide was recently approved for marketing in the U.S. for the treatment of Tourette's syndrome (TS). TS is characterized by recurrent, involuntary motor movements and vocal tics, and is believed to be due to neurochemical dysfunction. Pimozide's
J Rathbone et al.
The Cochrane database of systematic reviews, (3)(3), CD001949-CD001949 (2007-07-20)
Pimozide, formulated in the 1960s, continues to be marketed for the care of people with schizophrenia or related psychoses such as delusional disorder. It has been associated with cardiotoxicity and sudden unexplained deaths. Electrocardiogram monitoring is now required before and
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)