914460
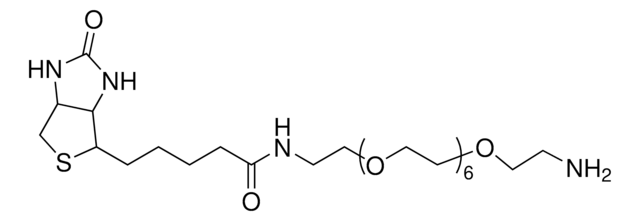
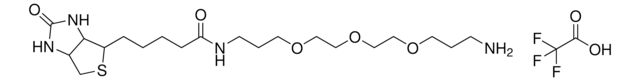
1-Biotinyl-3,6-dioxa-8-octaneamine hydrochloride
≥95%
Synonym(s):
1-Biotinyl-3,6-dioxa-8-octaneamine hydrochloride, N-(2-(2-(2-aminoethoxy)ethoxy)ethyl)-5-(2-oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamide hydrochloride, N-{2-[2-(2- Aminoethoxy)ethoxy]ethoxy}biotinamide hydrochloride, Amine-terminated biotin linker, Biotin-DOOA HCl, Biotinylation reagent
About This Item
Recommended Products
Application
Automate your Biotin tagging with Synple Automated Synthesis Platform (SYNPLE-SC002)
Other Notes
Caged Cyclopropenes with Improved Tetrazine Ligation Kinetics
A Unified Framework for the Incorporation of Bioorthogonal Compound Exposure Probes within Biological Compartments
A Chemical Probe for Protein Crotonylation
One-Step Selective Exoenzymatic Labeling (SEEL) Strategy for the Biotinylation and Identification of Glycoproteins of Living Cells
related product
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
JAN Code
914460-50MG:
914460-VAR:
914460-BULK:
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![O-(2-Aminoethyl)-O′-[2-(biotinylamino)ethyl]octaethylene glycol ≥95% (oligomer uniformity)](/deepweb/assets/sigmaaldrich/product/structures/300/798/e23e92b4-9ae0-4a8d-a37e-d57a8195a30e/640/e23e92b4-9ae0-4a8d-a37e-d57a8195a30e.png)