38292
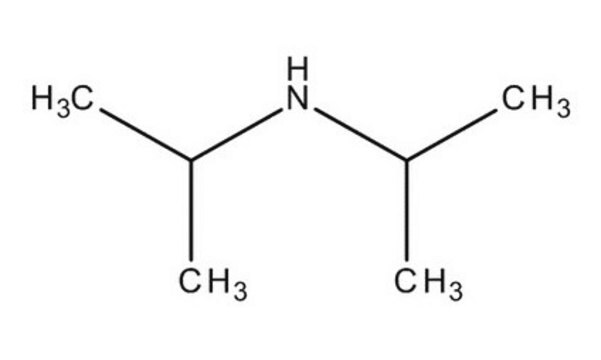
Diisopropylamine
analytical standard
Sinonimo/i:
DIPA
About This Item
Prodotti consigliati
Grado
analytical standard
Livello qualitativo
Densità del vapore
3.5 (vs air)
Tensione di vapore
50 mmHg ( 20 °C)
Saggio
≥99.5% (GC)
Temp. autoaccensione
599 °F
Durata
limited shelf life, expiry date on the label
Limite di esplosione
8.5 %
tecniche
HPLC: suitable
gas chromatography (GC): suitable
Indice di rifrazione
n20/D 1.392 (lit.)
n20/D 1.392
P. eboll.
84 °C (lit.)
Punto di fusione
−61 °C (lit.)
Solubilità
H2O: soluble 110 g/L at 25 °C (completely)
Densità
0.722 g/mL at 25 °C (lit.)
applicazioni
environmental
Formato
neat
Stringa SMILE
CC(C)NC(C)C
InChI
1S/C6H15N/c1-5(2)7-6(3)4/h5-7H,1-4H3
UAOMVDZJSHZZME-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Applicazioni
Prodotti consigliati
Avvertenze
Danger
Indicazioni di pericolo
Classi di pericolo
Acute Tox. 3 Inhalation - Acute Tox. 4 Oral - Aquatic Chronic 3 - Eye Dam. 1 - Flam. Liq. 2 - Skin Corr. 1B - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
3 - Flammable liquids
Classe di pericolosità dell'acqua (WGK)
WGK 2
Punto d’infiammabilità (°F)
7.8 °F - closed cup
Punto d’infiammabilità (°C)
-13.45 °C - closed cup
Dispositivi di protezione individuale
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.