424447
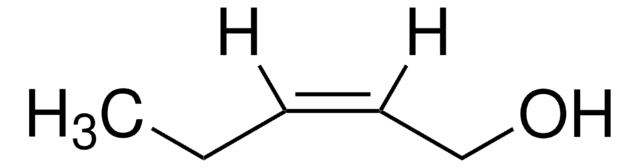
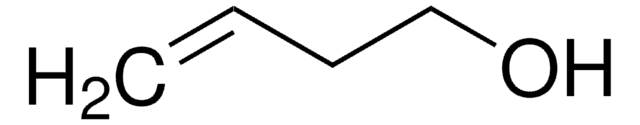
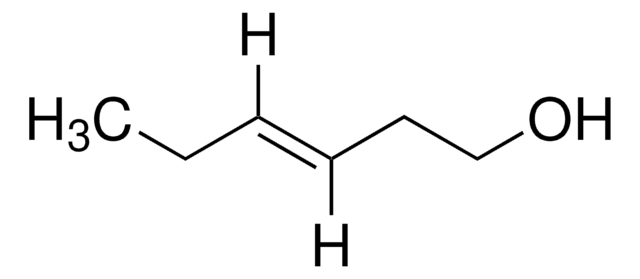
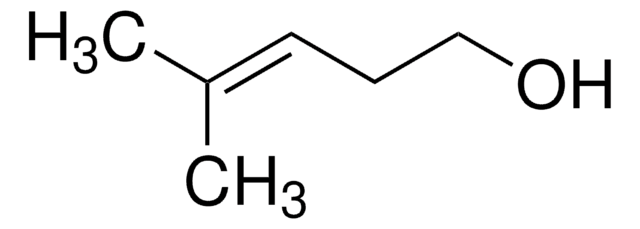
trans-2-Penten-1-ol
95%
Sinonimo/i:
(2E)-2-Penten-1-ol, (E)-Pent-2-en-1-ol, trans-2-Pentenol
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
C2H5CH=CHCH2OH
Numero CAS:
Peso molecolare:
86.13
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Saggio
95%
Stato
liquid
Indice di rifrazione
n20/D 1.434 (lit.)
P. ebollizione
139-139.5 °C (lit.)
Densità
0.847 g/mL at 25 °C (lit.)
Gruppo funzionale
hydroxyl
Stringa SMILE
CC\C=C\CO
InChI
1S/C5H10O/c1-2-3-4-5-6/h3-4,6H,2,5H2,1H3/b4-3+
BTSIZIIPFNVMHF-ONEGZZNKSA-N
Categorie correlate
Descrizione generale
trans-2-Penten-1-ol is an allyl alcohol. It is one of the volatile compounds found in olive oil, cashew apple juice and fermented cucumber brines. The rate constants and product ion distributions of its reaction with H3O+, NO+ and O2.+ ions have been studied using selected ion flow tube (SIFT).
Applicazioni
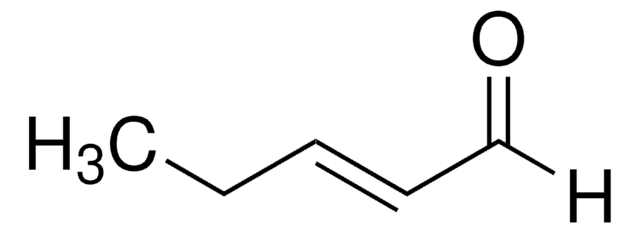
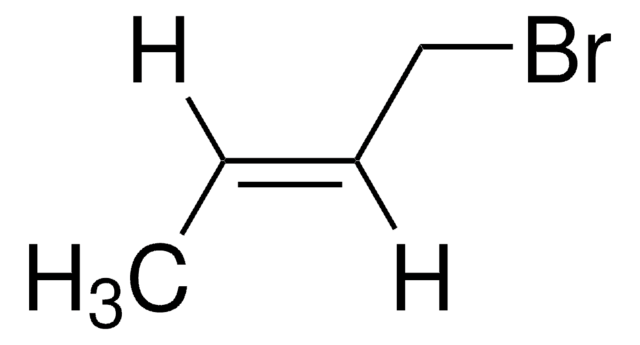
trans-2-Penten-1-ol may be used in the synthesis of the following:
- leustroducsin B
- trichloroacetimidate
- (E)-2,3,3′-trifluoro-4-(2-(trans-4-pentylcyclohexyl)ethyl)-4′-(pent-2-enyloxy)biphenyl
- trans-1-bromo-2-pentene
- trans-1-chloro-2-pentene
Avvertenze
Warning
Indicazioni di pericolo
Classi di pericolo
Flam. Liq. 3
Codice della classe di stoccaggio
3 - Flammable liquids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
118.4 °F - closed cup
Punto d’infiammabilità (°C)
48 °C - closed cup
Dispositivi di protezione individuale
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Kazuyuki Miyashita et al.
The Journal of organic chemistry, 73(14), 5360-5370 (2008-06-14)
Leustroducsin B was synthesized via a convergent route based on division of the leustroducsin molecule into three segments A, B, and C. Two coupling reactions (Julia coupling reaction and Nozaki-Hiyama-Kishi (NHK) reaction) were employed for coupling of segments A and
Suzanne D Johanningsmeier et al.
Journal of food science, 76(1), C168-C177 (2011-05-04)
A nontargeted, comprehensive 2-dimensional gas chromatography-time-of-flight mass spectrometry (GC×GC-TOFMS) method was developed for the analysis of fermented cucumber volatiles before and after anaerobic spoilage. Volatile compounds extracted by solid-phase microextraction were separated on a polyethylene glycol 1st-dimension column and 14%
Biogeneration of volatile compounds in virgin olive oil: their evolution in relation to malaxation time.
Angerosa F, et al.
Journal of Agricultural and Food Chemistry, 46(8), 2940-2944 (1998)
A selected ion flow tube study of the reactions of H3O+, NO+ and O2.+ with a series of C5, C6 and C8 unsaturated biogenic alcohols.
Schoon N, et al.
International Journal of Mass Spectrometry, 263(2-3), 127-136 (2007)
Stereoregulated synthesis of unsaturated compounds Communication 9. Stereochemistry of the reactions of aldehydes with ?, ?-unsaturated triphenylphosphonium ylides [alkylidenetriphenylphosphoranes].
Bergel'son LD, et al.
Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 15(3), 468-473 (1966)
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.