412562
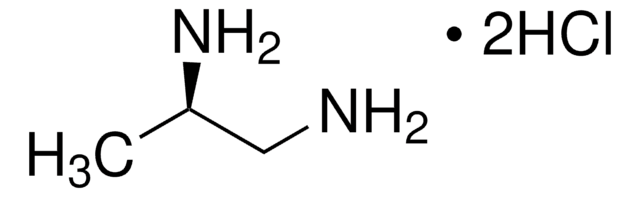
(S)-(−)-1,2-Diaminopropane dihydrochloride
99%
Sinonimo/i:
(S)-(−)-Propylenediamine dihydrochloride, (S)-1,2-Propanediamine dihydrochloride
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
CH3CH(NH2)CH2NH2·2HCl
Numero CAS:
Peso molecolare:
147.05
Beilstein:
5740936
Numero MDL:
Codice UNSPSC:
12352116
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Saggio
99%
Attività ottica
[α]22/D −4°, c = 20 in H2O
Punto di fusione
227-229 °C (lit.)
Gruppo funzionale
amine
Stringa SMILE
Cl.Cl.C[C@H](N)CN
InChI
1S/C3H10N2.2ClH/c1-3(5)2-4;;/h3H,2,4-5H2,1H3;2*1H/t3-;;/m0../s1
AEIAMRMQKCPGJR-QTNFYWBSSA-N
Categorie correlate
Applicazioni
(S)-(-)-1,2-Diaminopropane dihydrochloride may be used in the preparation of following chiral imidazoline derivatives, which show moderate α-adrenergic blocking activity:
- (S)-(-)-4-methyl-2-(1-naphthylmethyl)imidazoline hydrochloride
- (S)-(-)-2-benzyl-4-methylimidazoline picrate
- (S )-(-)-2-[(2,6-dichlorophenyl)imino]-4-methylimidazolidine hydrochloride
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Stereochemical studies of adrenergic drugs. Optically active derivatives of imidazolines.
Miller DD, et al.
Journal of Medicinal Chemistry, 19(12), 1382-1384 (1976)
Optically active derivatives of imidazolines: a-Adrenergic blocking properties.
Hsu FL, et al.
Journal of Medicinal Chemistry, 23(11), 1232-1235 (1980)
Chiral cyanide-bridged Mn(II)Mn(III) ferrimagnets, [Mn(II)(HL)(H2O)][Mn(III)(CN)6].2H2O (L = S- or R-1,2-diaminopropane): syntheses, structures, and magnetic behaviors.
Wakako Kaneko et al.
Journal of the American Chemical Society, 129(2), 248-249 (2007-01-11)
Keisuke Maruyoshi et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 15(7), 1618-1626 (2009-01-09)
Endogenous polyamines, represented by putrescine, spermidine, and spermine, are known to exert their physiological functions by interacting with polyanionic biomolecules such as DNA, RNA, adenosine triphosphate (ATP), and phospholipids. Very few examples of conformation analysis have been reported for these
S A M Fathi et al.
Journal of hazardous materials, 164(1), 133-137 (2008-09-10)
Bis(5-bromo-2-hydroxybenzaldehyde)-1,2-propanediimine is synthesized by the reaction of 5-bromo-2-hydroxybenzaldehyde and 1,2-diaminopropane in ethanol. This ligand is used as a modifier of octadecyl silica disks for preconcentration of trace amounts of copper(II) ions, followed by nitric acid elution and flame atomic absorption
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.