265004
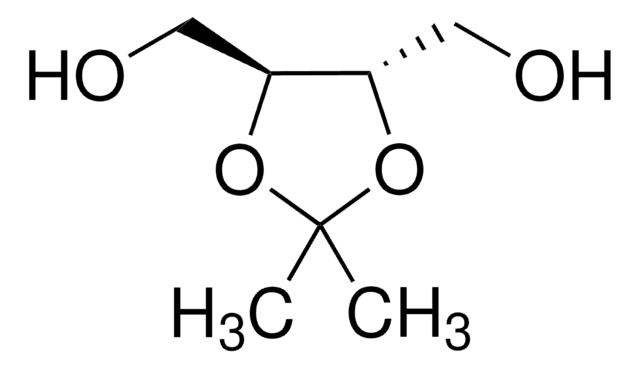
(4R,5R)-2,2-Dimethyl-α,α,α′,α′-tetraphenyldioxolane-4,5-dimethanol
97%
Sinonimo/i:
(−)-2,3-O-Isopropylidene-1,1,4,4-tetraphenyl-L-threitol, (−)-trans-α,α′-(2,2-Dimethyl-1,3-dioxolane-4,5-diyl)bis(diphenylmethanol), (4R,5R)-4,5-Bis(diphenylhydroxymethyl)-2,2-dimethyldioxolane, 1,1,4,4-Tetraphenyl-2,3-O-isopropylidene-L-threitol, TADDOL
About This Item
Prodotti consigliati
Saggio
97%
Attività ottica
[α]19/D −62.6°, c = 1 in chloroform
Punto di fusione
193-195 °C (lit.)
Gruppo funzionale
ether
hydroxyl
ketal
phenyl
Stringa SMILE
CC1(C)O[C@H]([C@@H](O1)C(O)(c2ccccc2)c3ccccc3)C(O)(c4ccccc4)c5ccccc5
InChI
1S/C31H30O4/c1-29(2)34-27(30(32,23-15-7-3-8-16-23)24-17-9-4-10-18-24)28(35-29)31(33,25-19-11-5-12-20-25)26-21-13-6-14-22-26/h3-22,27-28,32-33H,1-2H3/t27-,28-/m1/s1
OWVIRVJQDVCGQX-VSGBNLITSA-N
Applicazioni
Catalyst involved in synthesis of cyclopropylamines via addition reactions of Grignard reagents to amides
Reactant or reagent involved in:
- Enantioswitching of catalytic asymmetric hydroboration
- Synthesis of derivative ligands for asymmetric hydroformylation of alkenes
- Amide-directed catalytic asymmetric hydroboration of trisubstituted alkenes
- Addition of deactivated alkyl Grignard reagents to aldehydes
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Certificati d'analisi (COA)
Non trovi la versione di tuo interesse?
Se hai bisogno di una versione specifica, puoi cercare il certificato tramite il numero di lotto.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Articoli
The chiral auxiliaries TADDOLs (α,α,α,α-tetraaryl-1,3-dioxolane-4,5- dimethanols) developed by Seebach's group have found numerous applications in asymmetric synthesis ranging from utilization as stoichiometric chiral reagents or in Lewis acid mediated reactions, to roles in catalytic hydrogenation and stereoregular metathesis polymerization.
Apart from numerous examples using TADDOLs in metal-catalyzed asymmetric reactions, Rawal recently reported that TADDOLs could be used as Brønsted acid organocatalysts in highly stereoselective hetero-Diels–Alder reactions.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.

![[Ir(dFCF3ppy)2-(5,5’-dCF3bpy)]PF6 ≥95%](/deepweb/assets/sigmaaldrich/product/structures/422/901/e00f3148-fb86-4f94-9e79-21d064c3f327/640/e00f3148-fb86-4f94-9e79-21d064c3f327.png)