195030
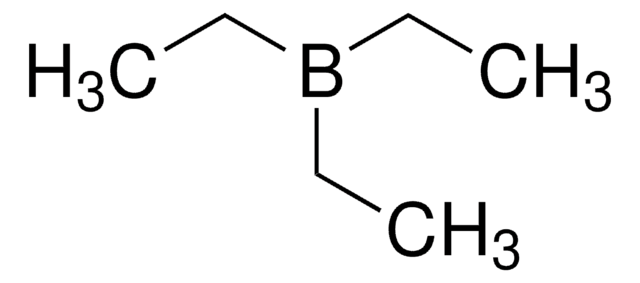
Triethylborane solution
1.0 M in hexanes
Sinonimo/i:
Triethylboron
About This Item
Prodotti consigliati
Stato
liquid
Impiego in reazioni chimiche
reagent type: reductant
Concentrazione
1.0 M in hexanes
Densità
0.675 g/mL at 25 °C
Stringa SMILE
CCB(CC)CC
InChI
1S/C6H15B/c1-4-7(5-2)6-3/h4-6H2,1-3H3
LALRXNPLTWZJIJ-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Applicazioni
- Allylation of aldehydes
- Decarboxylative C-C bond cleavage reactions
- Rhenium hydride / boron Lewis acid cocatalysis of alkene hydrogenations
- Regioselective hydroxyalkylation of unsaturated oxime ethers
Reactant for radical reductions of alkyl bromides with N-heterocyclic carbene boranes
Reactant for synthesis of tetramethylammonium trialkylphenylborate salts with oxidation potential
- As a radical initiator and terminator of free-radical reactions in aqueous media.(1)
- To synthesize polymers such as poly(2-substituted-1-propenylene)s by reacting with 2-substituted allylic arsonium ylides.(2)
Avvertenze
Danger
Indicazioni di pericolo
Classi di pericolo
Aquatic Chronic 2 - Asp. Tox. 1 - Eye Dam. 1 - Flam. Liq. 2 - Repr. 2 - Skin Corr. 1A - STOT RE 1 Inhalation - STOT SE 3
Organi bersaglio
Central nervous system, Nervous system
Codice della classe di stoccaggio
4.3 - Hazardous materials which set free flammable gases upon contact with water
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
-32.8 °F
Punto d’infiammabilità (°C)
-36 °C
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.