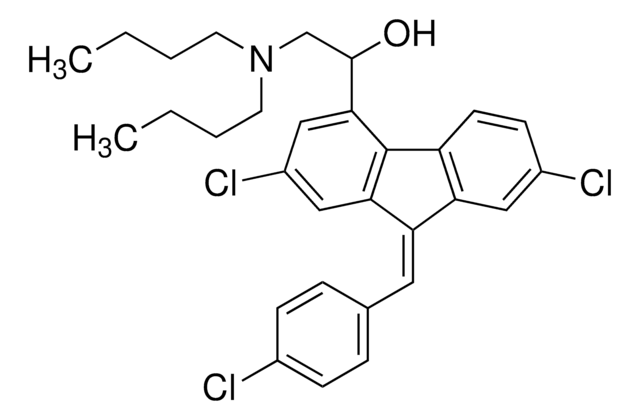
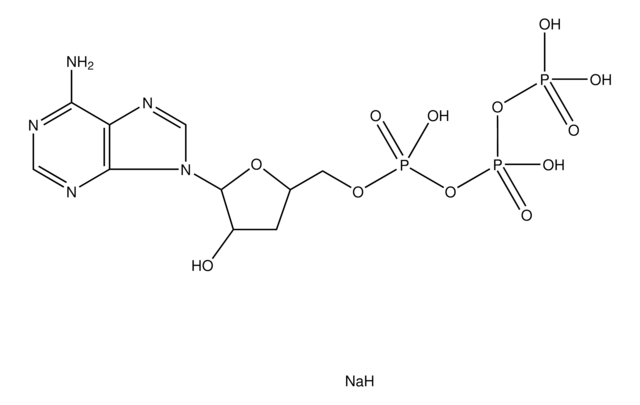
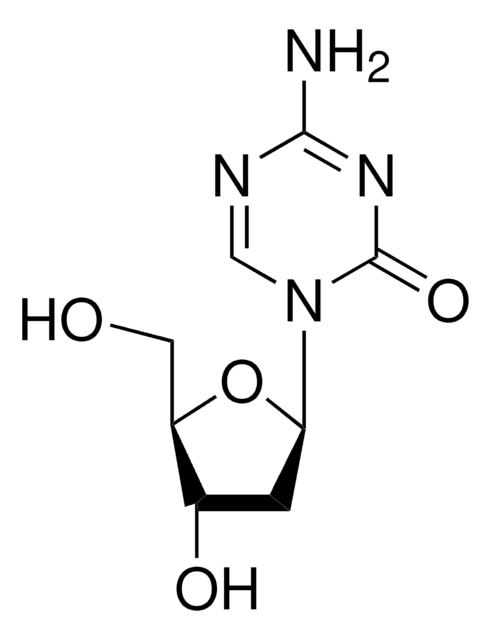
推荐产品
品質等級
化驗
≥98% (HPLC)
形狀
powder
顏色
white
溶解度
DMSO: >10 mg/mL
起源
Sanofi Aventis
儲存溫度
2-8°C
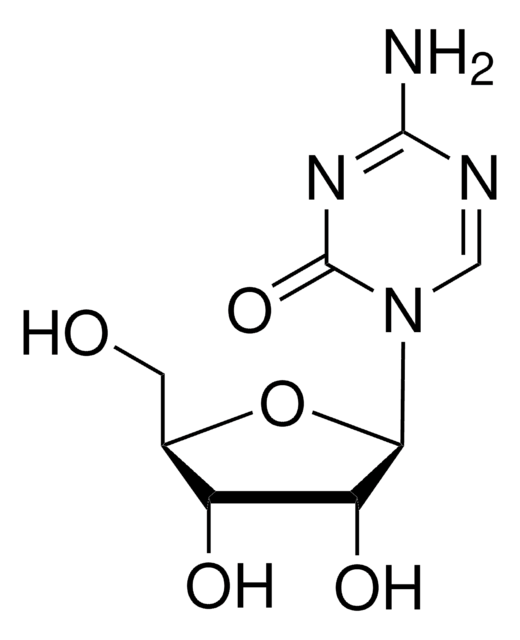
SMILES 字串
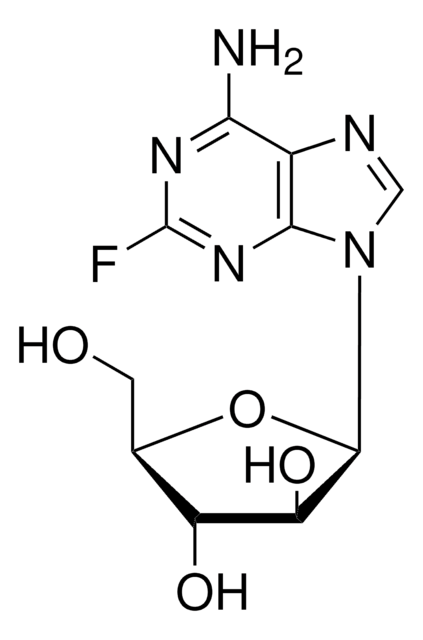
Nc1nc(Cl)nc2n(cnc12)[C@@H]3O[C@H](CO)[C@@H](O)[C@@H]3F
InChI
1S/C10H11ClFN5O3/c11-10-15-7(13)5-8(16-10)17(2-14-5)9-4(12)6(19)3(1-18)20-9/h2-4,6,9,18-19H,1H2,(H2,13,15,16)/t3-,4+,6-,9-/m1/s1
InChI 密鑰
WDDPHFBMKLOVOX-AYQXTPAHSA-N
基因資訊
human ... POLA1(5422) , POLA2(23649) , POLD1(5424) , POLD2(5425) , POLD3(10714) , POLD4(57804) , POLE(5426) , POLE2(5427) , POLE3(54107) , PRIM1(5557) , PRIM2(5558) , RRM1(6240) , RRM2(6241) , RRM2B(50484)
應用
生化/生理作用
特點和優勢
訊號詞
Warning
危險分類
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Carc. 2 - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
水污染物質分類(WGK)
WGK 2
個人防護裝備
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
其他客户在看
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门