推荐产品
一般說明
Aminopeptidase I from Streptomyces griseus is a thermostable enzyme with Glu131 and Tyr246 as key active site residues.
應用
Aminopeptidase I from Streptomyces griseus has been used:
- to test the biochar exposure effect on the enzyme activity
- in circular dichroism (CD) spectroscopy studies
- as a positive control in p-nitroanilide degradation assay
生化/生理作用
Aminopeptidase I from S. griseus has a fairly broad specificity, being able to remove the N-terminal residue of most proteins, except where the penultimate residue is an imino acid. It contains two Zn2+ binding sites. Aminopeptidase I from S. griseus is inhibited by 1,10-phenanthroline and is activated six-fold by Ca2+, which also stabilizes it against heat inactivation. This monomeric zinc metalloprotein has an isoelectric point (pI) of 5.4.
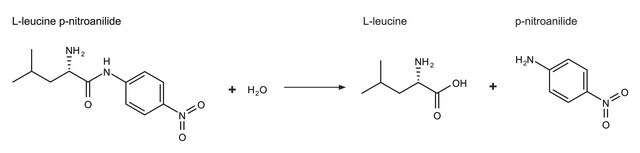
Aminopeptidase I may also be used as a reagent in the assay of endoprotease activities with a synthetic substrate in a two-stage assay. In the first stage, the endoprotease cleaves a peptide, such as Z-Y-X-Leu-p-nitroanilide, with the X, Y, and Z residues being chosen according to the specificity of the endoprotease.
包裝
Package size based on protein content.
單位定義
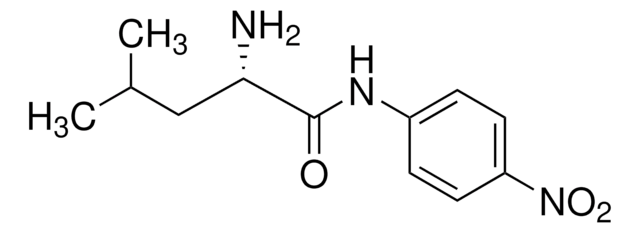
One unit will hydrolyze 1.0 μmole of L-leucine-p-nitroanilide to L-leucine and p-nitroaniline per min at pH 8.0, 25 °C and 3.0 mM substrate concentration.
外觀
Contains calcium acetate
準備報告
Reconstitute in 20 mM tricine, pH 8.0, with 0.05% bovine serum albumin. Dilute the enzyme with the reconstitution buffer to 0.15-0.3 U/mL for a working concentration. Solutions should be prepared fresh prior to use.
其他說明
Endopeptidase contaminant: Not more than: 0.01 U/mg protein (as μmole tyrosine equivalent per min released from casein.)
訊號詞
Danger
危險分類
Eye Irrit. 2 - Resp. Sens. 1 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 1
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, type N95 (US)
其他客户在看
[Autophagy related genes in yeast, S. cerevisiae].
Yoshinori Ohsumi
Tanpakushitsu kakusan koso. Protein, nucleic acid, enzyme, 51(10 Suppl), 1453-1456 (2006-08-23)
Indig, F.E., et al.
Febs Letters, 225, 237-237 (1989)
Taras Y Nazarko et al.
Autophagy, 1(1), 37-45 (2006-07-29)
Yarrowia lipolytica was recently introduced as a new model organism to study peroxisome degradation in yeasts. Transfer of Y. lipolytica cells from oleate/ethylamine to glucose/ammonium chloride medium leads to selective macroautophagy of peroxisomes. To decipher the molecular mechanisms of macropexophagy
A Spungin et al.
European journal of biochemistry, 183(2), 471-477 (1989-08-01)
A heat-stable aminopeptidase with an N-terminal Ala-Pro-Asp-Ile-Pro-Leu sequence has been purified from Streptomyces griseus by heat treatment followed by gel-exclusion and anion-exchange chromatographic procedures. The enzyme is a monomeric zinc metalloenzyme showing an apparent molecular mass of 33 kDa by
Congcong He et al.
Molecular biology of the cell, 19(12), 5506-5516 (2008-10-03)
Autophagy is the degradation of a cell's own components within lysosomes (or the analogous yeast vacuole), and its malfunction contributes to a variety of human diseases. Atg9 is the sole integral membrane protein required in formation of the initial sequestering
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门