所有图片(3)
About This Item
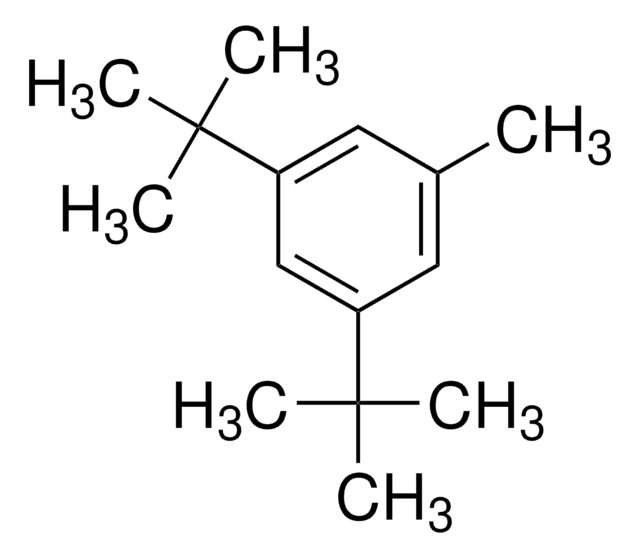
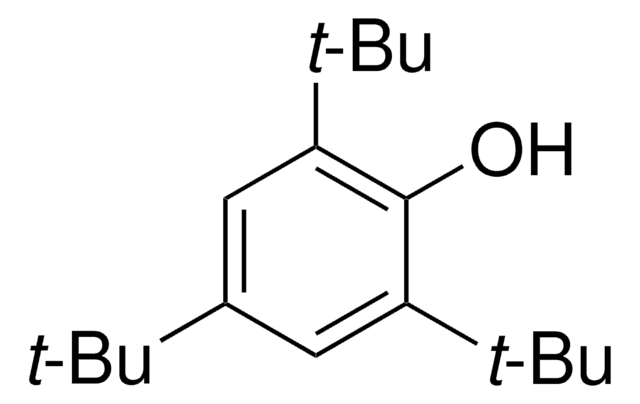
线性分子式:
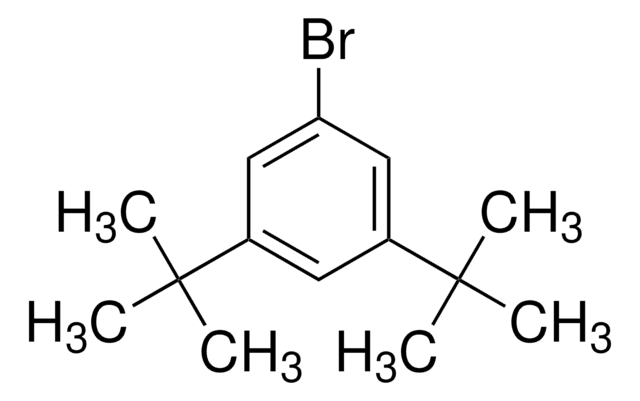
[(CH3)3C]3C6H2Br
CAS号:
分子量:
325.33
Beilstein:
1913257
MDL號碼:
分類程式碼代碼:
12352100
PubChem物質ID:
NACRES:
NA.22
推荐产品
化驗
97%
mp
168-173 °C (lit.)
SMILES 字串
CC(C)(C)c1cc(c(Br)c(c1)C(C)(C)C)C(C)(C)C
InChI
1S/C18H29Br/c1-16(2,3)12-10-13(17(4,5)6)15(19)14(11-12)18(7,8)9/h10-11H,1-9H3
InChI 密鑰
JOKZWHPYNRDCOA-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
1-Bromo-2,4,6-tri-tert-butylbenzene (2,4,6-tri-tert-butylbromobenzene) is a hindered aryl bromide. 1-Bromo-2,4,6-tri-tert-butylbenzene on reaction with phenylboronic acid yields α,α-dimethyl-β-phenyl hydrostyrene.
應用
1-Bromo-2,4,6-tri-tert-butylbenzene was used in the synthesis of bulky biarylphosphine ligand. This ligand was reported to participate in the Pd-catalyzed C-O cross-coupling of a wide range of aryl halides and phenols under milder conditions. It was used to investigate the effect on oligomerization of increased steric bulk in dimethylindium(III) chalcogenolates. It may be used to form α,α-dimethyl-β-phenyl hydrostyrene by reacting with phenylboronic acid.
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
其他客户在看
The first Cu-and amine-free Sonogashira-type cross-coupling in the C-6-alkynylation of protected 2'-deoxyadenosine.
Ngassa FN, et al.
Tetrahedron, 65(21), 4085-4091 (2009)
Luca Salvi et al.
Organic letters, 14(1), 170-173 (2011-12-21)
A new bulky biarylphosphine ligand (L8) has been developed that allows the Pd-catalyzed C-O cross-coupling of a wide range of aryl halides and phenols under milder conditions than previously possible. A direct correlation between the size of the ligand substituents
Palladium(0)-catalyzed intermolecular amination of unactivated C(sp³)-H bonds.
Jun Pan et al.
Angewandte Chemie (International ed. in English), 50(37), 8647-8651 (2011-08-04)
Glen G Briand et al.
Dalton transactions (Cambridge, England : 2003), 39(16), 3833-3841 (2010-04-08)
The effect on oligomerization of increased steric bulk in dimethylindium(III) chalcogenolates (Me(2)InER') (E = O, S, Se) has been examined. The facile reaction of Me(3)In with a series of phenols, thiophenols and selenophenols afforded the compounds [Me(2)InO(C(6)H(5))](2) (1), [Me(2)InO(2,6-Me(2)C(6)H(3))](2) (2)
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门