C109800
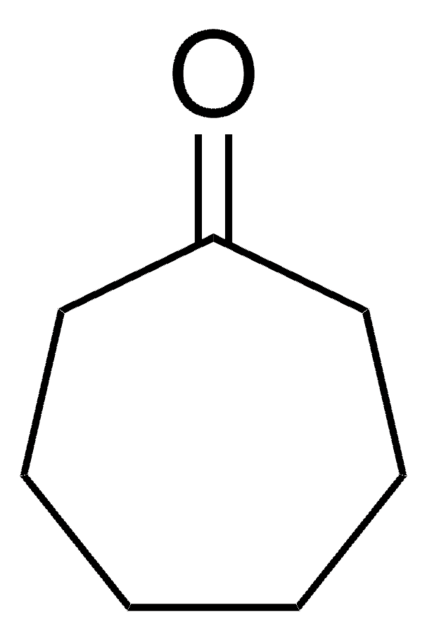
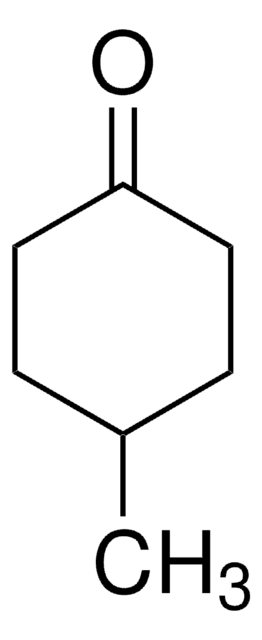
Cyclooctanone
98%
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
C8H14(=O)
Numero CAS:
Peso molecolare:
126.20
Beilstein:
1280738
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
98%
Stato
crystals
P. ebollizione
195-197 °C (lit.)
Punto di fusione
32-41 °C (lit.)
Densità
0.958 g/mL at 25 °C (lit.)
Stringa SMILE
O=C1CCCCCCC1
InChI
1S/C8H14O/c9-8-6-4-2-1-3-5-7-8/h1-7H2
IIRFCWANHMSDCG-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Avvertenze
Danger
Indicazioni di pericolo
Classi di pericolo
Eye Dam. 1 - Skin Corr. 1B
Codice della classe di stoccaggio
8A - Combustible corrosive hazardous materials
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
165.2 °F
Punto d’infiammabilità (°C)
74 °C
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Vishwakarma Singh et al.
The Journal of organic chemistry, 70(3), 973-981 (2005-01-29)
A new and efficient synthesis of a variety of highly embellished bicyclooctenones having an endo-vinyl moiety and their sigmatropic shifts in ground and excited states leading to a stereoselective route to substituted cis-decalins and diquinane frameworks have been described. Functionalized
M E Krafft et al.
The Journal of organic chemistry, 66(22), 7443-7448 (2001-10-30)
The total synthesis of asteriscanolide (1) has been achieved by taking advantage on an intermolecular Pauson-Khand cycloaddition and a ring-closing metathesis as key bond-forming transformations. The approach incorporates the cyclooctane stereogenic center prior to ring formation. Interestingly, the ring-closing metathesis
K Yamada et al.
Chemical & pharmaceutical bulletin, 45(12), 1898-1905 (1998-01-20)
Construction of the AB-ring system of the taxane framework via an A-ring annulation strategy was demonstrated by base-mediated intramolecular aldol reaction of (Z)-2,2-dimethyl-3-(1-methyl-2-oxopropylidene)cyclooctanone, affording the title compound, 1-hydroxy-8,11,11-trimethylbicyclo[5.3.1]undec-7-en-9-one. A cyclization precursor, the tetra-substituted (Z)-alkene, was prepared from the corresponding cyclooctanone
F E Harvey et al.
Brain research bulletin, 13(4), 541-547 (1984-10-01)
Female mice were reared in observation incubators from day 1 of life for three weeks. During that time they were continuously exposed to the odors of either cyclooctanone, adult male mouse urine or distilled water. The growth rate was temporarily
K Yamada et al.
Chemical & pharmaceutical bulletin, 45(12), 2113-2115 (1998-01-20)
Stereoselective syntheses of omega-(alpha-bromoketo) octanals and nonanal with oxygenated functions and formation of the corresponding eight-membered carbocyclic aldols by subsequent samarium(II)-mediated cyclization are demonstrated. Cyclooctenones deoxygenated at the C2 or C10 position in the taxane framework are prepared by dehydration
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.