A68300
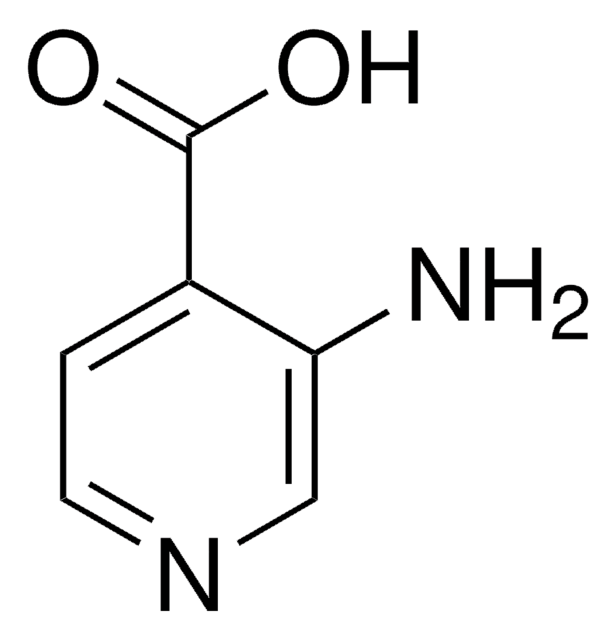
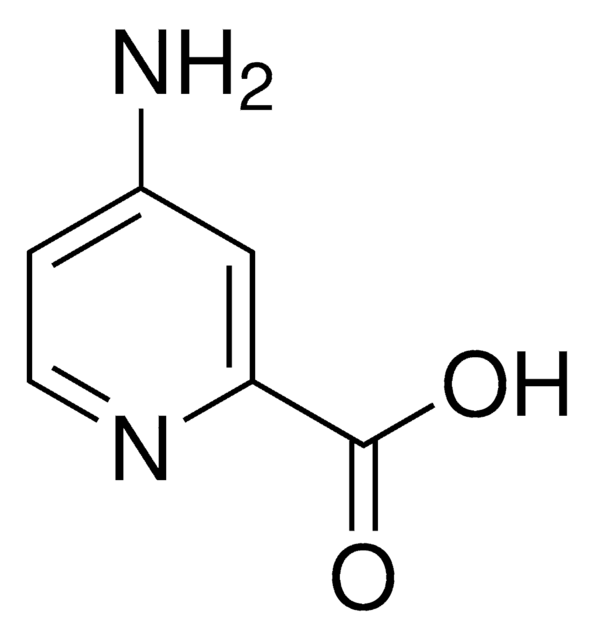
2-Aminopyridine-3-carboxylic acid
98%
Sinonimo/i:
2-Aminonicotinic acid, 2-Aminopyridine-3-carboxylic acid
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C6H6N2O2
Numero CAS:
Peso molecolare:
138.12
Beilstein:
119031
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
98%
Stato
powder
Punto di fusione
295-297 °C (dec.) (lit.)
Stringa SMILE
Nc1ncccc1C(O)=O
InChI
1S/C6H6N2O2/c7-5-4(6(9)10)2-1-3-8-5/h1-3H,(H2,7,8)(H,9,10)
KPIVDNYJNOPGBE-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
2-Aminopyridine-3-carboxylic acid can be used as:
- A ligand to prepare copper(II)-organic coordination compounds.
- A reactant to prepare pyrido[2′,1′:2,3]imidazo[4,5-c]isoquinolines by reacting with trimethylsilyl cyanide and phthalaldehyde.
- A reactant to synthesize organo-soluble and thermally stable poly(thiourea-amide-imide) polymers.
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Y Ohba et al.
Chemical & pharmaceutical bulletin, 48(8), 1236-1238 (2000-08-26)
To develop a sensitive and selective chemiluminometric method for the determination of methylglyoxal, we used 2-aminonicotinic acid as the chemiluminescence derivatization reagent. 2-Aminonicotinic acid reacts with methylglyoxal in an acidic solution at 37 degrees C for 4 h and gave
Synthesis, characterization, spectroscopic and electrochemical investigation of 2-aminopyridine-3-carboxylic acid copper (II) complexes with diimine
Srivastava AK, et al.
Chemical Data Collections, 24, 100272-100272 (2019)
Ronald Bartzatt
Drugs in R&D, 8(6), 363-372 (2007-10-30)
Nitrogen mustard (N-mustard) compounds are considered important anticancer drugs. Various transporting agents have been utilised to carry N-mustard groups including coumarins, amides, polyaromatic molecules and cycloalkyl structures. N-mustards act as bifunctional alkylating agents that induce cross-linking within DNA strands and
A J Dobson et al.
Acta crystallographica. Section C, Crystal structure communications, 53 ( Pt 10), 1427-1429 (1997-11-18)
2-Aminonicotinic acid, C6H6N2O2, crystallized in the centrosymmetric space group P2(1)/c in the zwitterionic form. Intermolecular N--H...O hydrogen bonds with N...O distances of 2.652 (2) and 2.807 (2) A link molecules into two sets of zigzag chains propagating along the b
Synthesis of pyrido [2′, 1′: 2, 3] imidazo [4, 5-c] isoquinolines via a one-pot, three-component reaction
Maleki A and Rezayan AH
Tetrahedron Letters, 55(10), 1848-1850 (2014)
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.