905291
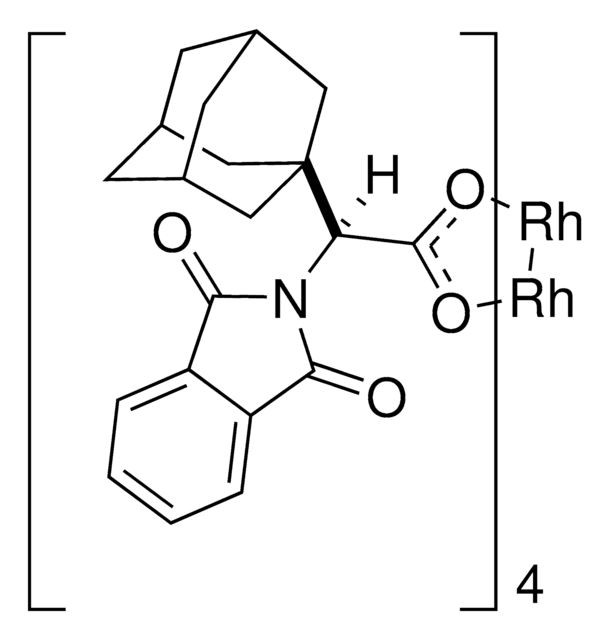
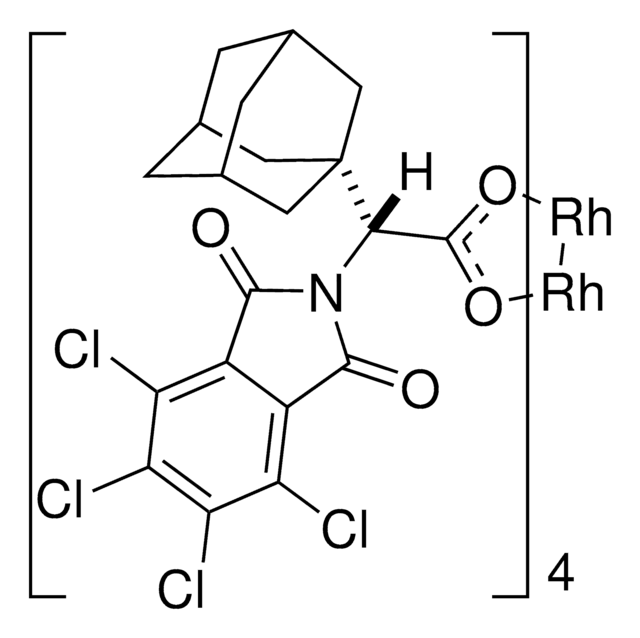
Rh2(R-BTPCP)4
Sinonimo/i:
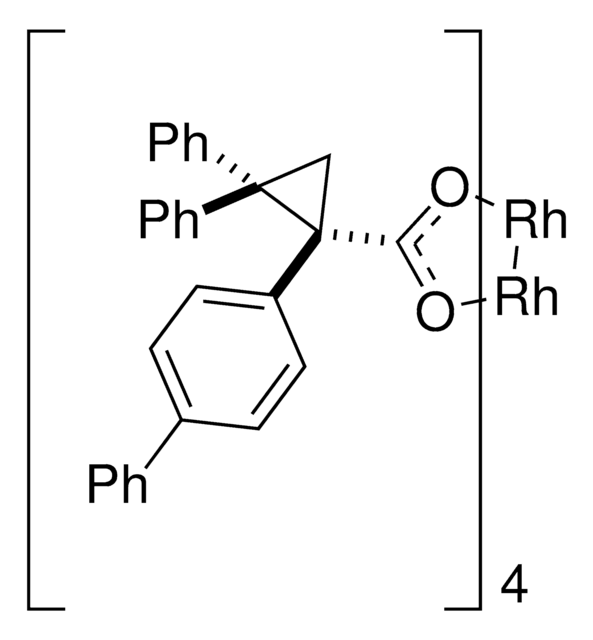
Davies dirhodium catalyst, Dirhodium tetrakis((S,R)-1-(4-bromophenyl)-2,2-diphenylcyclopropanecarboxylate), Dirhodium tetrakis[(R)-1-(4-bromophenyl)-2,2- diphenylcyclopropane carboxylate], Tetrakis[(R)-(-)-[(1R)-1-(4-bromophenyl)-2,2-diphenylcyclopropanecarboxylato]dirhodium(II), Tetrakis[(R)-1-(4-bromophenyl)-2,2- diphenylcyclopropane carboxylato]dirhodium(II)
About This Item
Prodotti consigliati
Stato
powder or crystals
Punto di fusione
>300 °C
Applicazioni
Prodotti correlati
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Scegli una delle versioni più recenti:
Certificati d'analisi (COA)
Non trovi la versione di tuo interesse?
Se hai bisogno di una versione specifica, puoi cercare il certificato tramite il numero di lotto.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.![Bis[rhodium(α,α,α′,α′-tetramethyl-1,3-benzenedipropionic acid)] 95%](/deepweb/assets/sigmaaldrich/product/structures/102/178/d1171a49-0358-406b-8b32-04324dbf9c02/640/d1171a49-0358-406b-8b32-04324dbf9c02.png)