335614
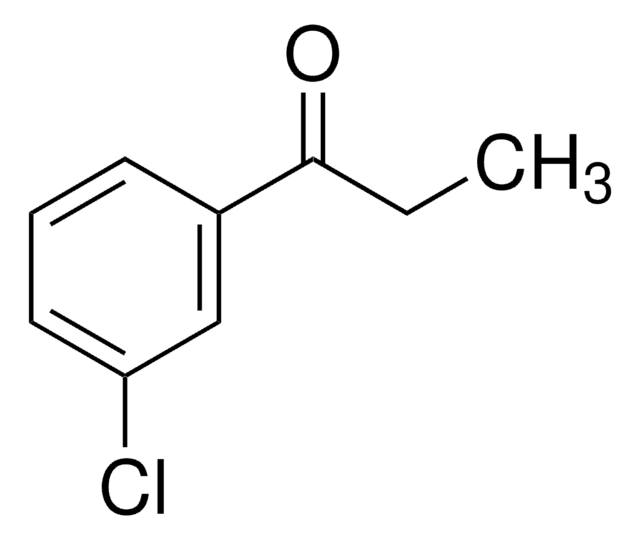
3-Chloropropiophenone
98%
Sinonimo/i:
β-Chloropropiophenone, 3-Chloro-1-phenyl-1-propanone
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
ClCH2CH2COC6H5
Numero CAS:
Peso molecolare:
168.62
Beilstein:
2043580
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
98%
Stato
solid
P. ebollizione
113-115 °C/4 mmHg (lit.)
Punto di fusione
48-50 °C (lit.)
Gruppo funzionale
chloro
ketone
phenyl
Stringa SMILE
ClCCC(=O)c1ccccc1
InChI
1S/C9H9ClO/c10-7-6-9(11)8-4-2-1-3-5-8/h1-5H,6-7H2
KTJRGPZVSKWRTJ-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
3-Chloropropiophenone was used in the asymmetric reduction of (S)-3-chloro-1-phenylpropanol using preheated Candida utilis cells immobilized in calcium alginate gel beads. It was also used in the synthesis of (R)-3-chloro-1-phenyl-1-propanol via asymmetric reduction using in-situ generated oxazaborolidine catalyst derived from (S)-α,α-diphenylprolinol.
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Asymmetric Synthesis of (R)-Fluoxetine: A Practical Approach Using Recyclable and in-situ Generated Oxazaborolidine Catalyst.
Padiya K, et al.
Chin. J. Chem., 27(6), 1137-1140 (2009)
Yang Gen-Sheng et al.
Biotechnology letters, 31(12), 1879-1883 (2009-07-28)
An efficient method for asymmetric reduction of (S)-3-chloro-1-phenylpropanol from 3-chloropropiophenone was developed using preheated Candida utilis cells immobilized in calcium alginate gel beads. Heating the immobilized cells (bead diameter 1.5 mm) at 45 degrees C for 50 min allowed the
Milada Šírová et al.
Journal of drug targeting, 25(9-10), 796-808 (2017-07-21)
Polymer carriers based on N-(2-hydroxypropyl)methacrylamide (HPMA) copolymers with incorporated organic nitrates as nitric oxide (NO) donors were designed with the aim to localise NO generation in solid tumours, thus highly increasing the enhanced permeability and retention (EPR) effect. The NO
Yu-Chang Liu et al.
Organic & biomolecular chemistry, 13(7), 2146-2152 (2014-12-23)
Styrene monooxygenase (SMO) can catalyze the kinetic resolution of secondary allylic alcohols to provide enantiopure glycidol derivatives. To overcome the low theoretical yield of kinetic resolution, we designed a one-pot two-step enzymatic cascade using prochiral α,β-unsaturated ketones as the substrates.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 335614-10G | 4061826740101 |
| 335614-50G | 4061831829433 |
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.