286338
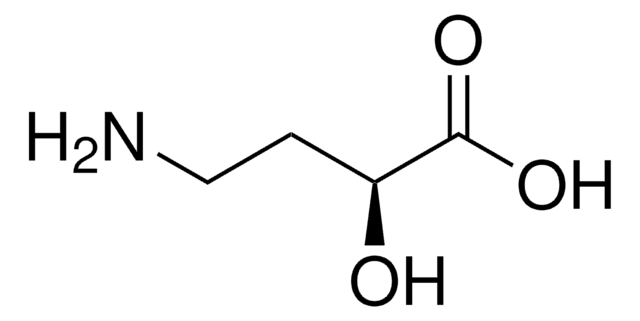
DL-Isoserine
98%
Sinonimo/i:
(±)-3-Amino-2-hydroxypropionic acid
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
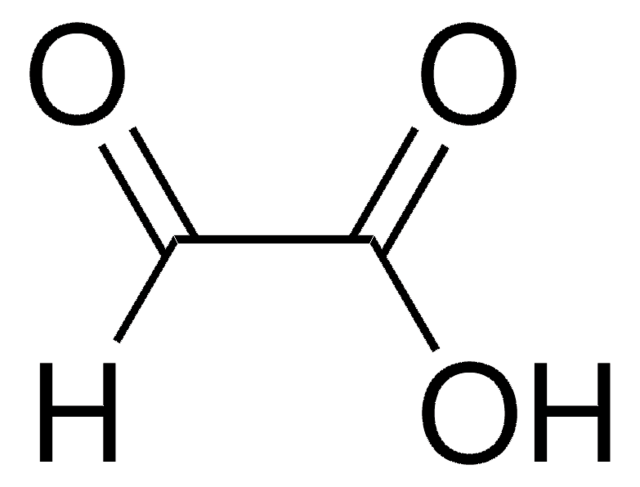
Formula condensata:
H2NCH2CH(OH)CO2H
Numero CAS:
Peso molecolare:
105.09
Beilstein:
1721413
Numero MDL:
Codice UNSPSC:
12352209
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Saggio
98%
Forma fisica
powder
Impiego in reazioni chimiche
reaction type: solution phase peptide synthesis
Punto di fusione
235 °C (dec.) (lit.)
applicazioni
peptide synthesis
Stringa SMILE
NCC(O)C(O)=O
InChI
1S/C3H7NO3/c4-1-2(5)3(6)7/h2,5H,1,4H2,(H,6,7)
BMYNFMYTOJXKLE-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Mei-Xiang Wang et al.
The Journal of organic chemistry, 70(7), 2439-2444 (2005-03-25)
[reaction: see text] Biotransformations of a number of differently substituted and configured oxiranecarbonitriles using Rhodococcus sp. AJ270, a microbial whole-cell catalyst that contains nitrile hydratase/amidase, were studied. While almost all trans-configured 3-aryl-2-methyloxiranecarbonitriles and 2,3-dimethyl-3-phenyloxiranecarbonitrile were efficiently hydrated by the action
Microbial resolution of 2-hydroxy-3-nitropropionic acid for synthesis of optically active isoserine.
Y Yasohara et al.
Bioscience, biotechnology, and biochemistry, 65(5), 1258-1260 (2001-07-07)
The biocatalytic stereoselective hydrolysis of 2-hydroxy-3-nitropropionic acid esters was studied. Forty enzymes and three hundred microorganism strains were examined for their ability to hydrolyze ethyl 2-hydroxy-3-nitropropionic acid. Nocardia globerula IFO13150 gave n-butyl (R)-2-hydroxy-3-nitropropionate with a 92% enantiomeric excess (ee) and
Jan Cz Dobrowolski et al.
Physical chemistry chemical physics : PCCP, 12(36), 10818-10830 (2010-07-10)
The IR low-temperature Ar and Kr matrix spectra of l-isoserine were registered for the first time and interpreted by means of the anharmonic DFT frequencies calculated at the B3LYP/aug-cc-pVTZ and B3LYP/aug-cc-pVDZ levels. 54 l-isoserine conformers were predicted to be stable
Alexander Titz et al.
Bioorganic & medicinal chemistry, 18(1), 19-27 (2009-12-02)
The selectin-leukocyte interaction is the initial event in the early inflammatory cascade. This interplay proceeds via the terminal tetrasaccharide sialyl Lewis(x) (sLe(x)), present on physiological selectin ligands and E- and P-selectins located on the endothelial surface. Blocking this process is
J Du et al.
Nucleosides & nucleotides, 17(1-3), 1-13 (1998-08-26)
Asymmetric synthesis of N-substituted oxazolidinyl nucleosides has been accomplished from L-isoserine, trans- and cis-Oxazolidine intermediates (4 and 5) were stereoselectively constructed from N-protected L-isoserine with a menthoxycarbonyl group by the condensation with benzoyloxy acetaldehyde dimethyl acetal in a ratio of
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.