272000
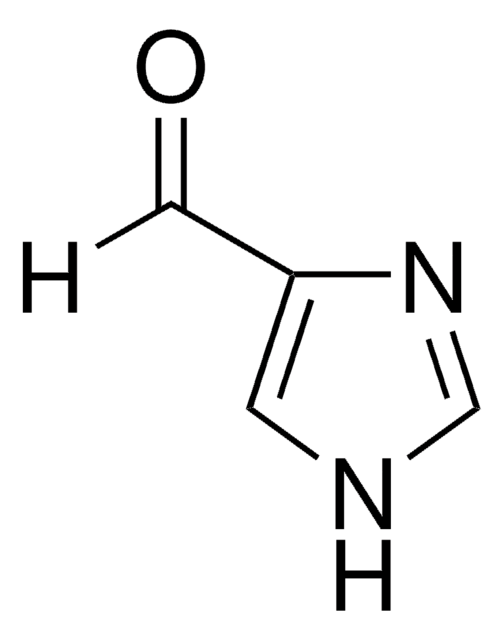
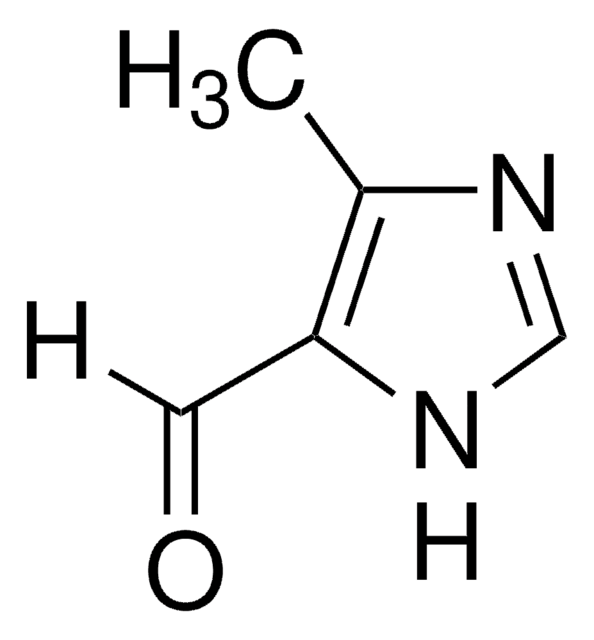
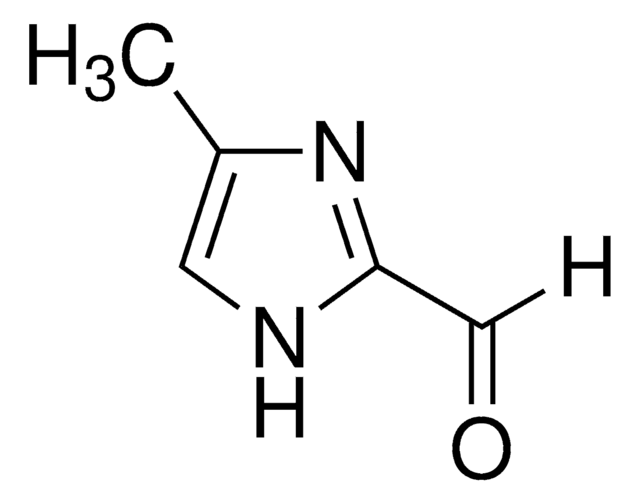
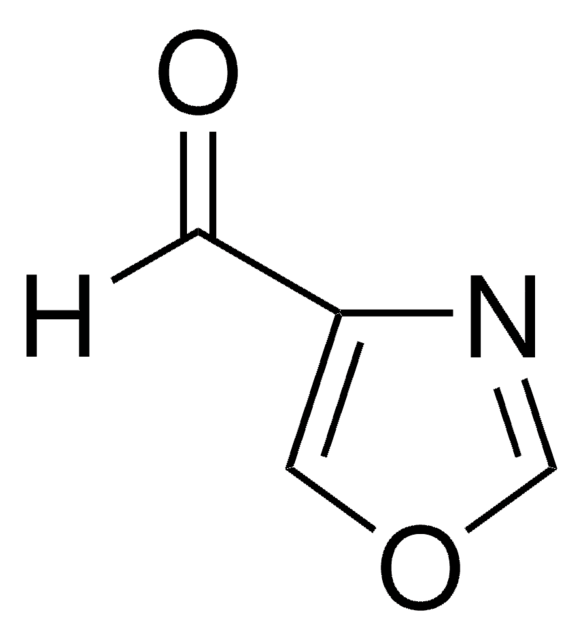
2-Imidazolecarboxaldehyde
97%
Sinonimo/i:
1H-Imidazole-2-carbaldehyde, 2-Formylimidazole, 2-Imidazolecarbaldehyde, 2-Imidazolylformaldehyde, Imidazole-2-formaldehyde
About This Item
Prodotti consigliati
Livello qualitativo
Saggio
97%
Punto di fusione
209 °C (dec.) (lit.)
Gruppo funzionale
aldehyde
Stringa SMILE
[H]C(=O)c1ncc[nH]1
InChI
1S/C4H4N2O/c7-3-4-5-1-2-6-4/h1-3H,(H,5,6)
XYHKNCXZYYTLRG-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 272000-100G | |
| 272000-1G | 4061826143995 |
| 272000-250MG | |
| 272000-5G | 4061826144008 |
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.