M9445
Matrix Metalloproteinase-2 from mouse
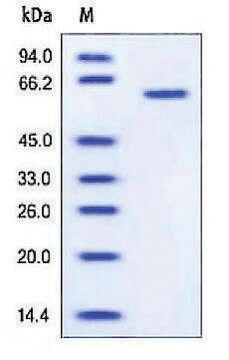
recombinant, expressed in NSO cells, >95% (SDS-PAGE), buffered aqueous glycerol solution
Sinônimo(s):
Collagenase Type IV 72 kDa, Gelatinase 72 kDa, Gelatinase A, MMP-2
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Produtos recomendados
recombinante
expressed in NSO cells
Nível de qualidade
Ensaio
>95% (SDS-PAGE)
Formulário
buffered aqueous glycerol solution
peso molecular
apparent mol wt ~72 kDa
Condições de expedição
wet ice
temperatura de armazenamento
−20°C
Informações sobre genes
mouse ... Mmp2(17390)
rat ... Mmp2(81686)
Descrição geral
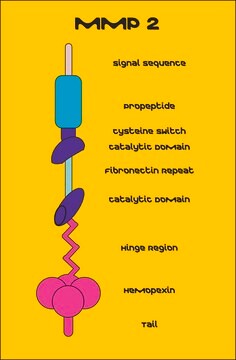
Matrix Metalloproteinase-2 (MMP-2) also known as gelatinase or type IV collagenase is a 72kDa protein. MMP-2 is a member of matrix metalloproteinase (MMP) family of enzymes. Basic structure of MMP2 contains signal peptide domain that targets the enzyme for secretion, the pro-peptide domain, which is removed when the enzyme is activated and the catalytic site containing gelatin-binding domain.
Especificidade
The amino acid sequences 1-662 of the proenzymes of MMP-2 are identical between mouse and rat.
Aplicação
Matrix metalloproteinase-2 (MMP2) human has been used as a standard in zymography to measure proteolytic activity of MMP-2.
Ações bioquímicas/fisiológicas
MMP-2 degrades general matrix components and may have a role in processes such as host defense, cell proliferation, and protein turnover as well as tissue remodeling.
Matrix Metalloproteinase-2 (MMP-2) cleaves gelatin, type IV, V, VII, X, and XI collagens, fibronectin, elastin, laminin, proteoglycans and a range of non extracellular matrix (ECM ) components. MMP-2 cleaves native type I collagen to N-terminal ¾ and C-terminal ¼ fragments identical to those generated by interstitial collagenases. MMP2 and MMP9 play an essential role in matrix degradation and they are implicated in the maintenance of neovascularization. In mice, deletion or inhibition of MMP2 protects against myocardial rupture.
forma física
Supplied as a 0.2 μm filtered solution of 25 mM Tris, pH 7.5, 5 mM calcium chloride, 75 mM sodium chloride, 0.025% Brij® 35 and 50% glycerol.
Nota de análise
The biological activity is measured by its ability to cleave a fluorogenic peptide sustrate.
Informações legais
Brij is a registered trademark of Croda International PLC
Código de classe de armazenamento
12 - Non Combustible Liquids
Classe de risco de água (WGK)
WGK 2
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, multi-purpose combination respirator cartridge (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Gelatinase A
Murphy, G. et al.
Handbook of Proteolytic Enzymes, 497-503 (2004)
INHIBITION OF GELATINASE ACTIVITY OF
MMP-2 AND MMP-9 BY EXTRACTS OF Bauhinia
ungulata L. Kamilla.
MMP-2 AND MMP-9 BY EXTRACTS OF Bauhinia
ungulata L. Kamilla.
Bioscience Journal, 31, 584-590 (2015)
Inhibition of Gelatinases by Vegetable Extracts
of the Species Tapirira guianensis
(Stick Pigeon).
of the Species Tapirira guianensis
(Stick Pigeon).
Longatti T R
British Journal of Pharmaceutical Research, 1(4), 133-140 (2011)
Immunohistochemical expression of MMP-14 and MMP-2, and MMP-2 activity during human ovarian follicular development.
Vos MC
Reproductive Biology and Endocrinology, 12:12 (2014)
P Reponen et al.
The Journal of biological chemistry, 267(11), 7856-7862 (1992-04-15)
We report the isolation of a cDNA clone providing the first and complete sequence of mouse 72-kDa type IV collagenase. The clone contains 2800 nucleotides with a 1986-nucleotide open reading frame coding for 662 amino acids. The amino acid sequence
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica