H5002
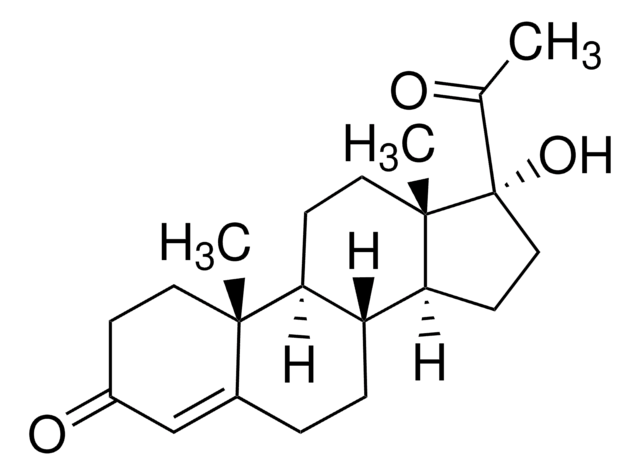
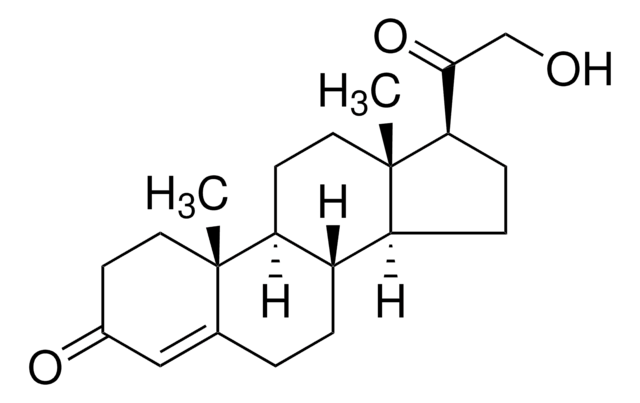
17α-Hydroxypregnenolone
Sinônimo(s):
3β,17α-Dihydroxy-5-pregnen-20-one, 5-Pregnene-3β,17α-diol-20-one
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula empírica (Notação de Hill):
C21H32O3
Número CAS:
Peso molecular:
332.48
Número CE:
Número MDL:
Código UNSPSC:
41116107
ID de substância PubChem:
NACRES:
NA.77
Produtos recomendados
cadeia de caracteres SMILES
C[C@]1(CC[C@H](O)C2)C2=CC[C@]3([H])[C@]1([H])CC[C@@]4(C)[C@@]3([H])CC[C@@]4(C(C)=O)O
InChI
1S/C21H32O3/c1-13(22)21(24)11-8-18-16-5-4-14-12-15(23)6-9-19(14,2)17(16)7-10-20(18,21)3/h4,15-18,23-24H,5-12H2,1-3H3
chave InChI
JERGUCIJOXJXHF-UHFFFAOYSA-N
Descrição geral
17α-hydroxypregnenolone is a derived from pregnenolone.
Aplicação
17α-hydroxypregnenolone has been used as a substrate for the enzyme 3β‐hydroxysteroid dehydrogenase (3β‐HSD) expressed in COS1 cells.
Ações bioquímicas/fisiológicas
17α-hydroxypregnenolone acts as a precursor for cortisol and sex steroids.
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Shogo Haraguchi et al.
Endocrinology, 153(2), 794-805 (2011-12-01)
7α-Hydroxypregnenolone (7α-OH PREG) is a newly identified bioactive neurosteroid stimulating locomotor activity in the brain of newt, a wild animal, which serves as an excellent model to investigate the biosynthesis and biological action of neurosteroids. Here, we show that acute
Kazuyoshi Tsutsui et al.
General and comparative endocrinology, 168(2), 275-279 (2010-02-09)
We now know that steroids can be synthesized de novo by the brain and the peripheral nervous system. Such steroids are called neurosteroids and de novo neurosteroidogenesis from cholesterol is a conserved property of vertebrate brains. Our studies over the
Kazuyoshi Tsutsui et al.
General and comparative endocrinology, 176(3), 440-447 (2011-12-06)
Seasonally-breeding amphibians have served as excellent animal models to investigate the biosynthesis and biological actions of neurosteroids. Previous studies have demonstrated that the brain of amphibians possesses key steroidogenic enzymes and produces pregnenolone, a precursor of steroid hormones, and other
Tamara S Hannon et al.
Journal of pediatric and adolescent gynecology, 25(1), 82-85 (2011-11-18)
Little is known about racial differences in androgen levels among obese children. The objective of this pilot study was to compare basal and stimulated androgen levels in a cross-sectional sample of obese black and white pubertal females. STUDY DESIGN, SETTING
Teppei Koyama et al.
Annals of the New York Academy of Sciences, 1163, 444-447 (2009-05-22)
We recently identified 7alpha-hydroxypregnenolone as a novel amphibian neurosteroid stimulating locomotor activity in newts. Because male newts show marked diurnal changes in locomotor activity, we hypothesized that 7alpha-hydroxypregnenolone may be a key factor for the induction of diurnal changes in
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica