F8512
Anti-Fibrinogen antibody produced in goat
whole antiserum
Sinônimo(s):
Anti-Fib2
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Produtos recomendados
fonte biológica
goat
conjugado
unconjugated
forma do anticorpo
whole antiserum
tipo de produto de anticorpo
primary antibodies
clone
polyclonal
contém
15 mM sodium azide
reatividade de espécies
human
técnica(s)
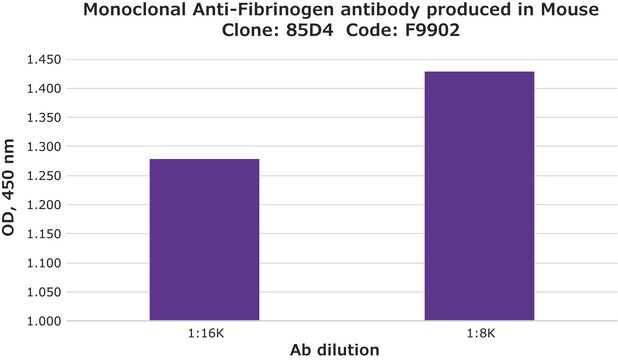
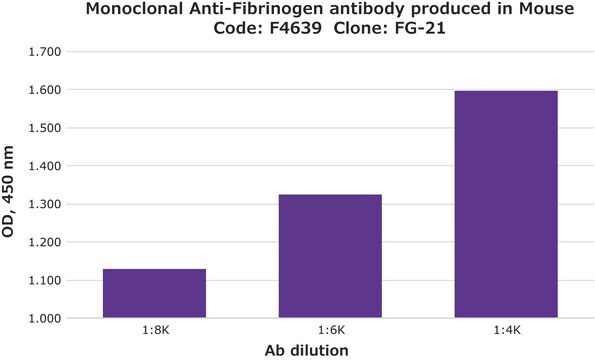
indirect ELISA: 1:10,000
quantitative precipitin assay: 2.0 mg/mL
nº de adesão UniProt
temperatura de armazenamento
−20°C
modificação pós-traducional do alvo
unmodified
Categorias relacionadas
Descrição geral
Fibrinogen is a thrombin-coagulable soluble plasma 340 kDa glycoprotein, composed of paired sets of three subunits i.e. α, β, γ. It plays a crucial role in protecting the vascular network against the loss of blood after tissue injury. Among three subunits, β and γ subunits contain one N-glycosylation site, which is occupied by a biantennary N-glycan. It contains three pairs of disulfide-bonded chains called α, β and γ which further folded into four structural domains: the D, E, connector, and the COOH-terminal region of the Aα chain.
The fibrinogen gene cluster consists of fibrinogen α, β and γ chains. It is localized on human chromosome locus 4q31.3−4q32.1.
Especificidade
The antiserum has been determined to be immunospecific for fibrinogen by immunoelectrophoresis versus human plasma and fibrinogen.
Imunogênio
human fibrinogen
Aplicação
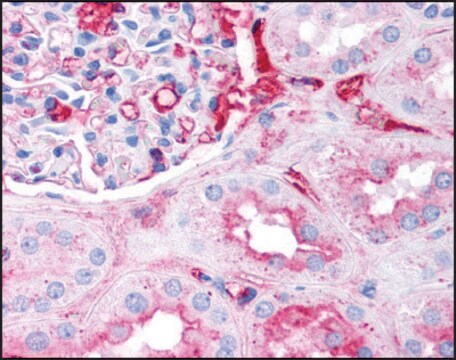
Anti-Fibrinogen antibody is suitable for immunostaining in fibrin deposition analysis of mouse livers and capturing antibodies in the sandwich ELISA. It is also suitable for indirect ELISA at a dilution of 1:10,000 and quantitative precipitin assay at 2.0mg/mL concentration.
Anti-Fibrinogen antibody produced in goat has been used in:
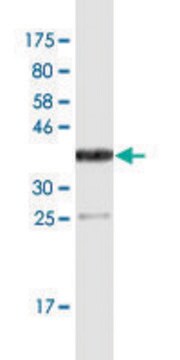
- western blotting detection in human colon adenocarcinoma cell line
- detecting fibrinogen in plasma
- immunoassay of human platelet free plasma (PFP)
Ações bioquímicas/fisiológicas
Plasmin attacks the Aα chain COOH domain to produce the heterogeneous fragment X. Multiple round of degradation ended with terminal digestion products−fragments D and E which represent the major globular domains in fibrinogen. Mutations in this gene lead to several disorders, including hypofibrinogenemia and afibrinogenemia.
Nota de preparo
treated to remove lipoproteins
Exoneração de responsabilidade
Unless otherwise stated in our catalog or other company documentation accompanying the product(s), our products are intended for research use only and are not to be used for any other purpose, which includes but is not limited to, unauthorized commercial uses, in vitro diagnostic uses, ex vivo or in vivo therapeutic uses or any type of consumption or application to humans or animals.
Não está encontrando o produto certo?
Experimente o nosso Ferramenta de seleção de produtos.
Código de classe de armazenamento
12 - Non Combustible Liquids
Classe de risco de água (WGK)
nwg
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Congenital afibrinogenaemia caused by uniparental isodisomy of chromosome 4 containing a novel 15-kb deletion involving fibrinogen Aalpha-chain gene.
Spena S, et al.
European Journal of Human Genetics, 12(11), 891-891 (2004)
Integrin alphavbeta6 mediates HT29-D4 cell adhesion to MMP-processed fibrinogen in the presence of Mn2+.
Fouchier F, et al.
European Journal of Cell Biology, 86(3), 143-160 (2007)
A quantitative immunoassay for lung cancer biomarker CIZ1b in patient plasma.
Coverley D, et al.
Clinical Biochemistry, 50(6), 336-343 (2017)
Platelet adhesion and plasma protein adsorption control of collagen surfaces by He+ ion implantation.
Kurotobi K, et al.
Nucl. Instrum. Methods Phys. Res. Sect. B, 206(6), 532-537 (2003)
N E Kirschbaum et al.
The Journal of biological chemistry, 265(23), 13669-13676 (1990-08-15)
The COOH-terminal portion of the A alpha chain of human fibrinogen is highly susceptible to proteolytic degradation. This property has prevented isolation of the COOH-terminal domain of fibrinogen for the direct investigation of its functional characteristics. Human fibrinogen was degraded
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica