D1446
Dexrazoxane
≥95% (HPLC)
Sinônimo(s):
(+)-(S)-4,4′-Propylenedi-2,6-piperazinedione, (S)-(+)-1,2-Bis(3,5-dioxopiperazin-1-yl)propane, Cardioxane, ICRF-187, NSC169780, Zinecard
About This Item
Produtos recomendados
Ensaio
≥95% (HPLC)
Formulário
powder
cor
white to off-white
solubilidade
DMSO: >20 mg/mL
originador
Johnson & Johnson
temperatura de armazenamento
room temp
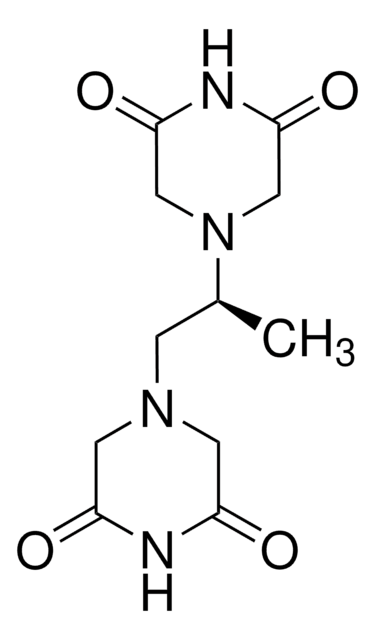
cadeia de caracteres SMILES
C[C@@H](CN1CC(=O)NC(=O)C1)N2CC(=O)NC(=O)C2
InChI
1S/C11H16N4O4/c1-7(15-5-10(18)13-11(19)6-15)2-14-3-8(16)12-9(17)4-14/h7H,2-6H2,1H3,(H,12,16,17)(H,13,18,19)/t7-/m0/s1
chave InChI
BMKDZUISNHGIBY-ZETCQYMHSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Descrição geral
Aplicação
Ações bioquímicas/fisiológicas
Características e benefícios
Palavra indicadora
Warning
Frases de perigo
Declarações de precaução
Classificações de perigo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órgãos-alvo
Respiratory system
Código de classe de armazenamento
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Conteúdo relacionado
Apoptosis, or programmed cell death (PCD), is a selective process for the removal of unnecessary, infected or transformed cells in various biological systems. As it plays a role in the homeostasis of multicellular organisms, apoptosis is tightly regulated through two principal pathways by a number of regulatory and effector molecules.
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica