N17602
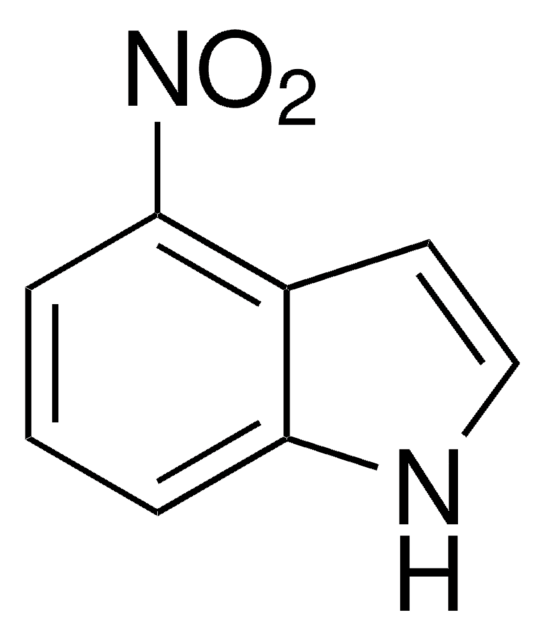
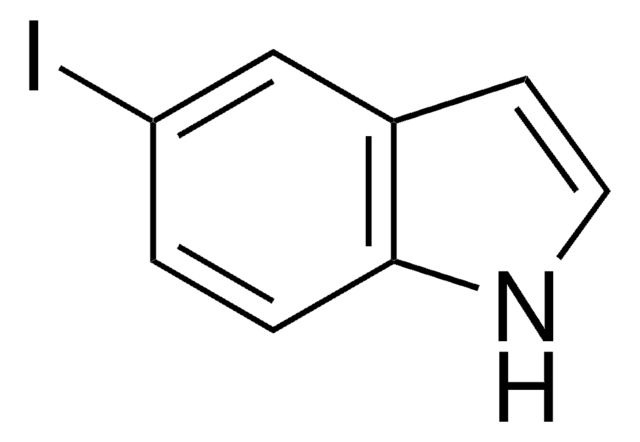
5-Nitroindole
98%
Sinônimo(s):
NSC 520594
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula empírica (Notação de Hill):
C8H6N2O2
Número CAS:
Peso molecular:
162.15
Beilstein:
383777
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Nível de qualidade
Ensaio
98%
Formulário
powder
pf
140-142 °C (lit.)
cadeia de caracteres SMILES
[O-][N+](=O)c1ccc2[nH]ccc2c1
InChI
1S/C8H6N2O2/c11-10(12)7-1-2-8-6(5-7)3-4-9-8/h1-5,9H
chave InChI
OZFPSOBLQZPIAV-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Aplicação
Reactant for preparation of:
- Pharmaceutically active 2-oxo-1-pyrrolidine analogues
- Tryptophan dioxygenase inhibitors pyridyl-ethenyl-indoles as potential anticancer immunomodulators
- Protein Kinase Inhibitors and antiproliferative agents
- Positive Allosteric Modulators of Metabotropic Glutamate Receptor 4 (mGlu4)
- Antifungal agents
- Cannabinoid receptor type 1 (CB1) antagonists
- Potential anticancer agents
- Potential antivascular agents
- Selective Anti-leukemic agents
- Anti human immunodeficiency virus subtype 1 (HIV-1) agents
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Acute Tox. 4 Oral - Eye Dam. 1
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
José Gallego et al.
Nucleic acids research, 35(9), 2904-2912 (2007-04-18)
Universal bases hybridize with all other natural DNA or RNA bases, and have applications in PCR and sequencing. We have analysed by nuclear magnetic resonance spectroscopy the structure and dynamics of three DNA oligonucleotides containing the universal base analogues 5-nitroindole
H Challa et al.
Organic letters, 1(10), 1639-1641 (2000-06-03)
[formula: see text] The syntheses of PNA oligomers containing potential ambiguous nucleobase analogues, namely 3-nitropyrrole and 5-nitroindole, have been accomplished. Hybridization properties of these PNAs with complementary oligodeoxynucleotides were evaluated by thermal denaturation experiments. Both novel residues exhibited little variation
P M Vallone et al.
Nucleic acids research, 27(17), 3589-3596 (1999-08-14)
Effects of the universal base 5-nitroindole on the thermodynamic stability of DNA hairpins having a 6 bp stem and four base loops were investigated by optical absorbance and differential scanning calorimetry techniques. Melting studies were conducted in buffer containing 115
Use of 5-nitroindole-2'-deoxyribose-5'-triphosphate for labelling and detection of oligonucleotides.
C L Smith et al.
Nucleosides & nucleotides, 17(1-3), 555-564 (1998-08-26)
The 5'-triphosphate of 5-nitroindole-2'-deoxyriboside has been shown to be a good substrate for terminal deoxynucleotidyl transferase (TdT). An antibody has been prepared for the detection of 5-nitroindole and has been used for the detection of 5-nitroindole tailed DNA both in
David Loakes et al.
Journal of the American Chemical Society, 131(41), 14827-14837 (2009-09-26)
Hydrophobic base analogues (HBAs) have shown great promise for the expansion of the chemical and coding potential of nucleic acids but are generally poor polymerase substrates. While extensive synthetic efforts have yielded examples of HBAs with favorable substrate properties, their
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica