G6104
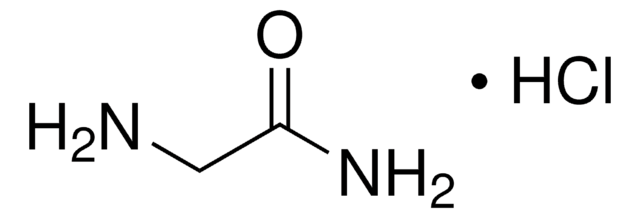
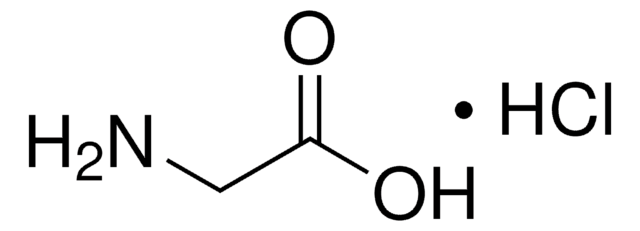
Glycinamide hydrochloride
98%
Sinônimo(s):
2-Aminoacetamide hydrochloride, Aminoacetamide hydrochloride, Glycine amide hydrochloride
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula linear:
NH2CH2CONH2 · HCl
Número CAS:
Peso molecular:
110.54
Beilstein:
3554199
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Nível de qualidade
Ensaio
98%
pf
204 °C (dec.) (lit.)
cadeia de caracteres SMILES
Cl.NCC(N)=O
InChI
1S/C2H6N2O.ClH/c3-1-2(4)5;/h1,3H2,(H2,4,5);1H
chave InChI
WKNMKGVLOWGGOU-UHFFFAOYSA-N
Aplicação
Buffer useful in the physiological pH range.
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Gottfried K Schroeder et al.
Biochemistry, 46(13), 4037-4044 (2007-03-14)
As a model for mechanistic comparison with peptidyl transfer within the ribosome, the reaction of aqueous glycinamide with N-formylphenylalanine trifluoroethyl ester (fPhe-TFE) represents an improvement over earlier model reactions involving Tris. The acidity of trifluoroethanol (pKa 12.4) resembles that of
Eric Loeser et al.
Analytical chemistry, 79(14), 5382-5391 (2007-05-29)
When mobile-phase salt content is increased, cationic analytes often show increased retention. This effect is generally attributed to chaotropic or ion pairing effects. However, a cation exclusion mechanism could explain the same effects. In this study, experimental conditions were manipulated
Brett C Bookser et al.
Journal of medicinal chemistry, 48(24), 7808-7820 (2005-11-24)
4-(Phenylamino)-5-phenyl-7-(5-deoxy-beta-D-ribofuranosyl)pyrrolo[2,3-d]pyrimidine 1 and related compounds known as "diaryltubercidin" analogues are potent inhibitors of adenosine kinase (AK) and are orally active in animal models of pain such as the rat formalin paw model (GP3269 ED50= 6.4 mg/kg). However, the utility of
Xiaodong Jia et al.
The Journal of organic chemistry, 78(18), 9450-9456 (2013-08-21)
A catalytic α-sp(3) C-H oxidation of peptides and glycine amides was achieved under radical cation salt catalysis in the presence of O2, producing a series of substituted quinolines. The scope of this reaction shows good functional group tolerance and high
Hong Zhao et al.
Bioconjugate chemistry, 17(2), 341-351 (2006-03-16)
The utility of PEGylation for improving therapeutic protein pharmacology would be substantially expanded if the authentic protein drugs could be regenerated in vivo. Diminution of kinetic constants of both enzymes and protein ligands are commonly encountered following permanent bioconjugation with
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica