G11950
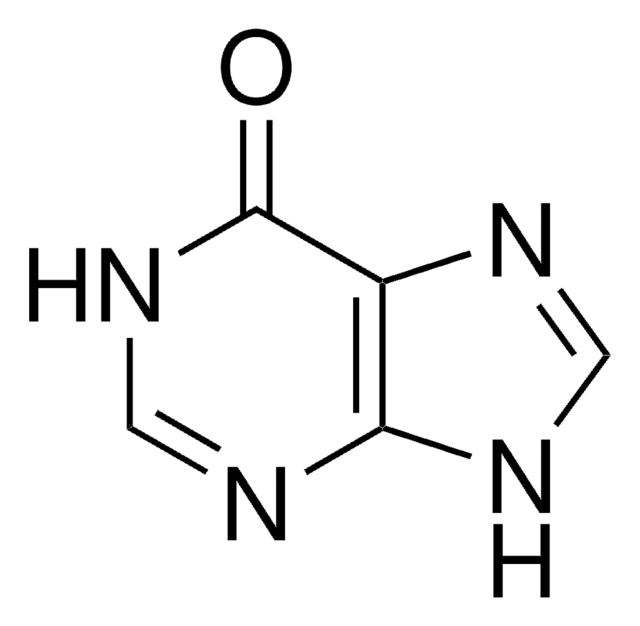
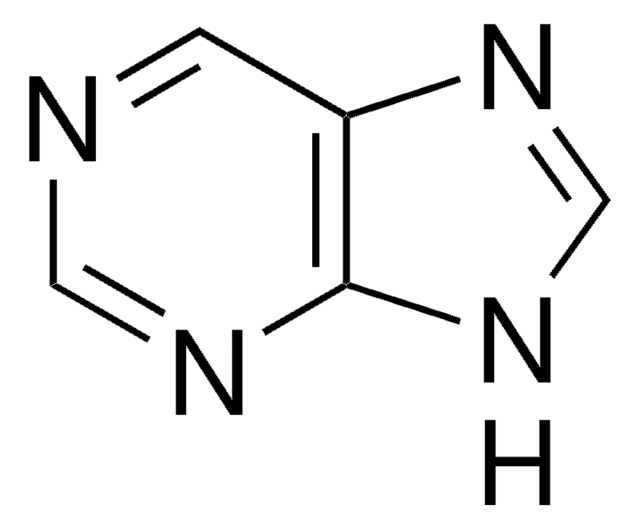
Guanine
98%
Sinônimo(s):
2-Amino-1,7-dihydro-6H-purin-6-one, 2-Amino-6-hydroxypurine, 2-Aminohypoxanthine
About This Item
Produtos recomendados
Nível de qualidade
Ensaio
98%
pf
>300 °C (lit.)
solubilidade
hydrochloric acid: soluble 5 M, clear to slightly hazy, colorless to faintly yellow-green
cadeia de caracteres SMILES
NC1=Nc2[nH]cnc2C(=O)N1
InChI
1S/C5H5N5O/c6-5-9-3-2(4(11)10-5)7-1-8-3/h1H,(H4,6,7,8,9,10,11)
chave InChI
UYTPUPDQBNUYGX-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Descrição geral
Aplicação
Palavra indicadora
Warning
Frases de perigo
Declarações de precaução
Classificações de perigo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órgãos-alvo
Respiratory system
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
dust mask type N95 (US), Eyeshields, Gloves
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Conteúdo relacionado
DISCOVER Bioactive Small Molecules for Neuroscience
Global Trade Item Number
| SKU | GTIN |
|---|---|
| G11950-100G | 4061838357830 |
| G11950-10G | 4061838671608 |
| G11950-25G | 4061833627983 |
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica