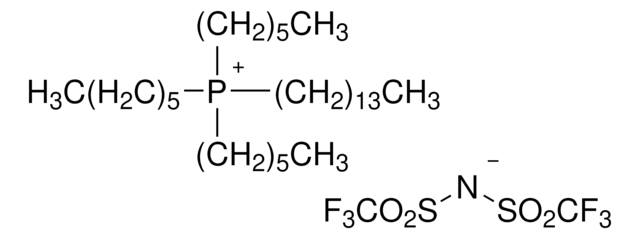727725
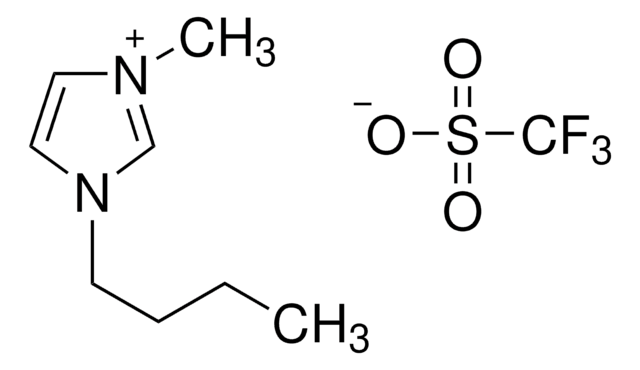
1-Butyl-1-methylpyrrolidinium trifluoromethanesulfonate
95%
Sinônimo(s):
BMPyrrOTf
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula empírica (Notação de Hill):
C10H20F3NO3S
Número CAS:
Peso molecular:
291.33
Beilstein:
9820503
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Ensaio
≥95.0% (T)
95%
Formulário
liquid
Impurezas
≤0.3% water
grupo funcional
fluoro
triflate
cadeia de caracteres SMILES
[O-]S(=O)(=O)C(F)(F)F.CCCC[N+]1(C)CCCC1
InChI
1S/C9H20N.CHF3O3S/c1-3-4-7-10(2)8-5-6-9-10;2-1(3,4)8(5,6)7/h3-9H2,1-2H3;(H,5,6,7)/q+1;/p-1
chave InChI
WZJDNKTZWIOOJE-UHFFFAOYSA-M
Aplicação
1-Butyl-1-methylpyrrolidinium trifluoromethanesulfonate ([BMPy][OTf]) is an ionic liquid that can be used as a solvent in:
([BMPy][OTf]) can also be used as an electrolyte in supercapacitor applications.
- Rhodium-catalyzed regioselective hydroformylation reactions.
- Direct asymmetric aldol condensation reaction.
- Desulfurization of fuels.
- Nucleophilic aromatic substitution reactions.
([BMPy][OTf]) can also be used as an electrolyte in supercapacitor applications.
Código de classe de armazenamento
10 - Combustible liquids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Hydroformylation of 1-hexene with rhodium in non-aqueous ionic liquids: how to design the solvent and the ligand to the reaction
Favre F, et al.
Chemical Communications (Cambridge, England), 1360-1361 (2001)
Andrzej Lewandowski et al.
Journal of solution chemistry, 42(2), 251-262 (2013-03-14)
The electrochemical behavior of cobaltocenium has been studied in a number of room temperature aprotic ionic liquids. Well defined, diffusion controlled, anodic and cathodic peaks were found for the Cc
An improved protocol for the direct asymmetric aldol reaction in ionic liquids, catalysed by onium ion-tagged prolines
Lombardo M, et al.
Advanced Synthesis & Catalysis, 349(11-12), 2061-2065 (2007)
Ionic liquids as designer solvents for nucleophilic aromatic substitutions
Newington I, et al.
Organic Letters, 9(25), 5247-5250 (2007)
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica