697230
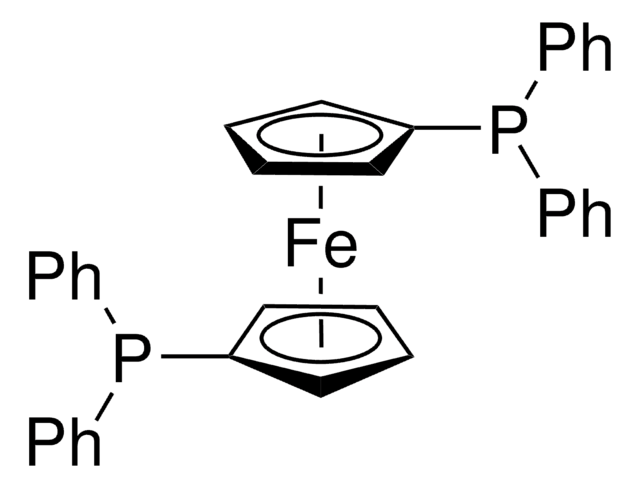
[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)
Sinônimo(s):
Pd(dppf)Cl2
About This Item
Produtos recomendados
forma
powder
Nível de qualidade
adequação da reação
core: palladium
reaction type: Cross Couplings
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: catalyst
reaction type: Buchwald-Hartwig Cross Coupling Reaction
pf
266-283 °C (lit.)
cadeia de caracteres SMILES
[Fe].Cl[Pd]Cl.[CH]1[CH][CH][C]([CH]1)P(c2ccccc2)c3ccccc3.[CH]4[CH][CH][C]([CH]4)P(c5ccccc5)c6ccccc6
InChI
1S/2C17H14P.2ClH.Fe.Pd/c2*1-3-9-15(10-4-1)18(17-13-7-8-14-17)16-11-5-2-6-12-16;;;;/h2*1-14H;2*1H;;/q;;;;;+2/p-2
chave InChI
JCWIWBWXCVGEAN-UHFFFAOYSA-L
Procurando produtos similares? Visita Guia de comparação de produtos
Aplicação
- Cross-coupling of sec-alkyl and n-alkyl Grignard reagents with high yield and selectivity.
- Suzuki coupling of aryl boronic esters with [11C]methyl iodide to form functionalized [11C]toluene derivatives.
- Kumada cross-coupling of 1,3,5-tribromobenzene with Grignard reagents to form star-shaped oligothiophenes.
produto relacionado
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Artigos
Tandem hydroboration Suzuki Coupling both intermolecular and intramolecular gave diverse alkyl substituted products dppf
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II), complex with dichloromethane](/deepweb/assets/sigmaaldrich/product/structures/825/986/4317978b-1256-4c82-ab74-6a6a3ef948b1/640/4317978b-1256-4c82-ab74-6a6a3ef948b1.png)




![[Pd(OAc)2]3 reagent grade, 98%](/deepweb/assets/sigmaaldrich/product/structures/508/249/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f/640/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f.png)