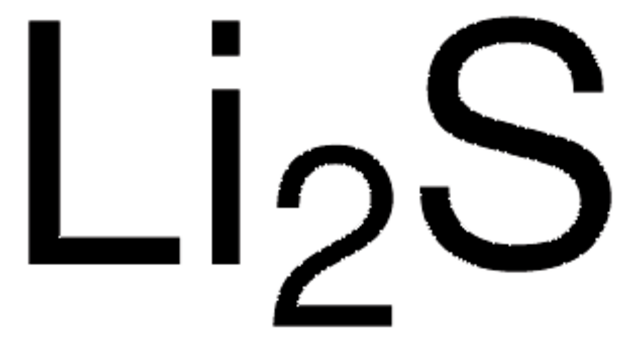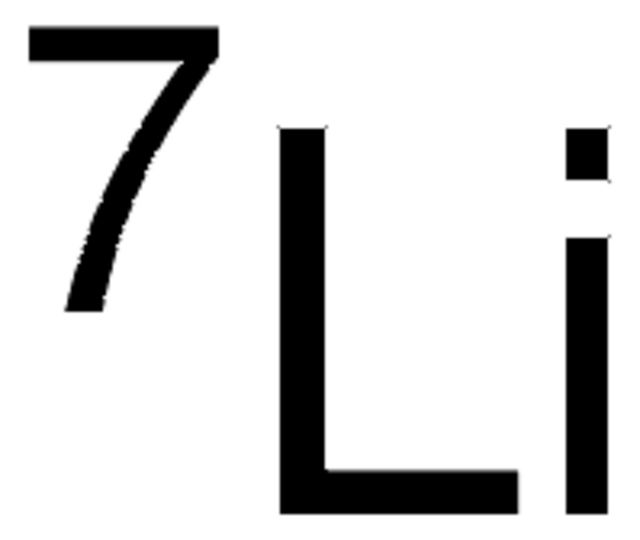499811
Lithium
granular, 99% trace metals basis
Sinônimo(s):
Lithium atom, Lithium element
About This Item
Produtos recomendados
Nível de qualidade
Ensaio
99% trace metals basis
Formulário
granular
adequação da reação
reagent type: reductant
resistividade
9.446 μΩ-cm, 20°C
p.e.
1342 °C (lit.)
pf
180 °C (lit.)
densidade
0.534 g/mL at 25 °C (lit.)
cadeia de caracteres SMILES
[Li]
InChI
1S/Li
chave InChI
WHXSMMKQMYFTQS-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Descrição geral
Aplicação
- A precursor to synthesize Li-based alloys such as LiAl and LiSi alloys, which are applicable as anode materials in the field of energy conversion and storage.
- A reducing agent in the reduction of zirconium oxide compounds in molten LiCl salt.
- As starting material to synthesize a reducing agent(1,4-bis(trimethylgermyl)-1.4-dihydropyrazine), for the fabrication of nickel metal films.
Palavra indicadora
Danger
Frases de perigo
Declarações de precaução
Classificações de perigo
Skin Corr. 1B - Water-react 1
Perigos de suplementos
Código de classe de armazenamento
4.3 - Hazardous materials which set free flammable gases upon contact with water
Classe de risco de água (WGK)
WGK 1
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica