About This Item
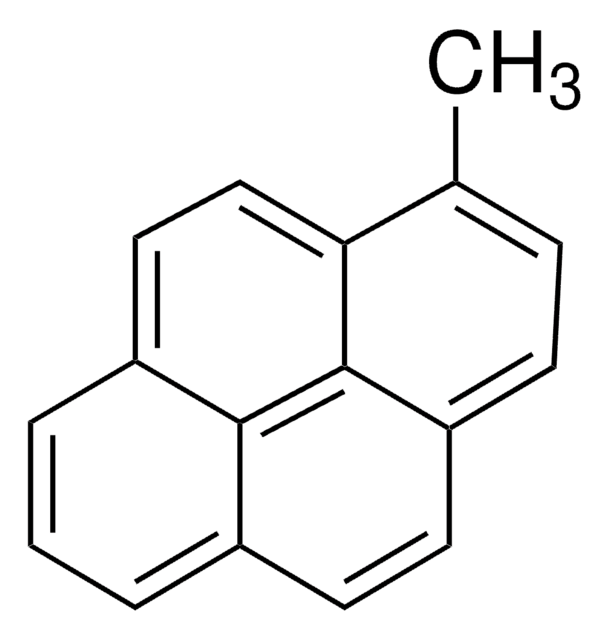
Fórmula empírica (Notação de Hill):
C18H12O
Número CAS:
Peso molecular:
244.29
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Ensaio
97%
Formulário
solid
pf
86-89 °C (lit.)
grupo funcional
ketone
cadeia de caracteres SMILES
CC(=O)c1ccc2ccc3cccc4ccc1c2c34
InChI
1S/C18H12O/c1-11(19)15-9-7-14-6-5-12-3-2-4-13-8-10-16(15)18(14)17(12)13/h2-10H,1H3
chave InChI
KCIJNJVCFPSUBQ-UHFFFAOYSA-N
Descrição geral
1-Acetylpyrene is a pyrene derivative. Its synthesis has been reported. Its phytophysical properties have been studied using absolute fluorescence quantum yield measurement and time-dependent density functional theory (TD-DFT) calculations. Its ability to interact with human cytochromes P450 2A13, 2A6, and 1B1 and enzyme inhibition has been reported. Its function as an environment-sensitive fluorophore has been investigated.
Aplicação
1-Acetylpyrene is suitable for use in a comparative study on the photoinitiating efficiency of pyrene, 1-acetylpyrene and 1-(bromoacetyl)pyrene for copolymerization of styrene with acrylonitrile. It may be used in the following studies:
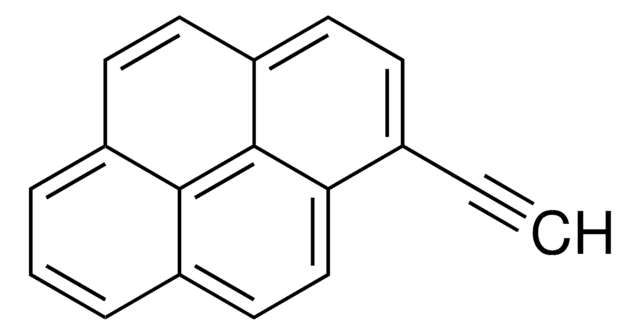
- As a starting material in the synthesis of ethynlypyrene. 1-(1-chlorovinyl)pyrene was also isolated during this reaction.
- As a starting material in the synthesis of substituted pyrene derivatives incorporated heterocyclic and sugar moieties.
- Synthesis of (E)-pyrene oxime ester conjugates of carboxylic acids.
- Synthesis of tertiary alcohols based on 1-acetylpyrene.
- Synthesis of (E)-N-[1-(pyren-1-yl)ethylidene]chrysene-2-amine.
- Synthesis of 3,3-di(methylsulfanyl)-1-(1-pyrenyl)-2-propen-1-one.
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
H Surya Prakash Rao et al.
Beilstein journal of organic chemistry, 3, 31-31 (2007-10-02)
The cycloaddition of the von Leusen's reagent (p-tolylsulfonyl)methyl isocyanide (TosMIC) to alpha-aroylketene dithioacetals (AKDTAs) in the presence of sodium hydride in THF at rt resulted in a facile synthesis of the 4-aroyl-3-methylsulfanyl-2-tosylpyrroles 3 in good yield along with a minor
Synthesis and study of film-forming properties and light sensitivity of 4-acyloxy-3-methoxy (ethoxy) phenylmethylidene-(chrysen-2-yl) amines.
Dikusar EA, et al.
Russ. J. Gen. Chem., 77(2), 278-281 (2007)
Nilanjana Chowdhury et al.
Photochemical & photobiological sciences : Official journal of the European Photochemistry Association and the European Society for Photobiology, 11(7), 1239-1250 (2012-05-09)
A new series of (E)-pyrene oxime ester conjugates of carboxylic acids including amino acids were synthesized by coupling with an environment sensitive fluorophore 1-acetylpyrene. (E)-Pyrene oxime esters exhibited strong fluorescence properties and interestingly their fluorescence properties were found to be
Fundamental photoluminescence properties of pyrene carbonyl compounds through absolute fluorescence quantum yield measurement and density functional theory.
Niko Y, et al.
Tetrahedron, 68(31), 6177-6185 (2012)
Synthesis, characterization and pharmacological investigations of some novel heterocyclic derivatives incorporating pyrene and sugar moieties.
Khalifa NM, et al.
Research on Chemical Intermediates, 40(4), 1565-1574 (2014)
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica