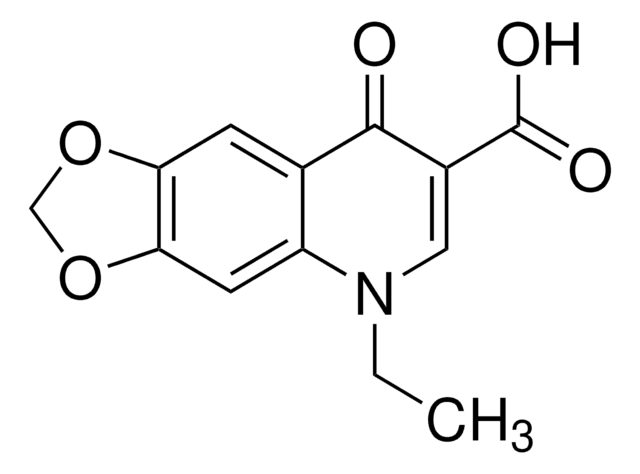
168696
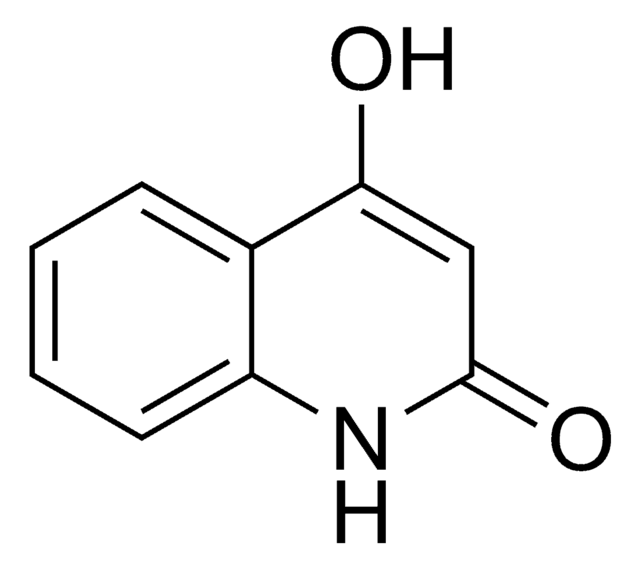
4-Hydroxy-1-methyl-2(1H)-quinolone
98%
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula empírica (Notação de Hill):
C10H9NO2
Número CAS:
Peso molecular:
175.18
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Ensaio
98%
forma
powder
pf
269-271 °C (lit.)
cadeia de caracteres SMILES
CN1C(=O)C=C(O)c2ccccc12
InChI
1S/C10H9NO2/c1-11-8-5-3-2-4-7(8)9(12)6-10(11)13/h2-6,12H,1H3
chave InChI
RTNPPPQVXREFKX-UHFFFAOYSA-N
Aplicação
4-Hydroxy-1-methyl-2(1H)-quinolone was used as nucleophile in electrochemical oxidation of catechols and the reaction was studied by cyclic voltammetry and controlled-potential coulometry. It was used in ceric ammonium nitrate-mediated oxidative cycloaddition of 1,3-dicarbonyls to conjugated compounds to yield substituted dihydrofurans, dihydrofurocoumarins, dihydrofuroquinolinones and dihydrofurophenalenones. It was used in the synthesis of :
- 3-(4-methoxybenzyl)-4-hydroxy-1-methylquinolinone
- 3-(benzo[d][1,3]dioxol-5-ylmethyl)-4-hydroxy-1-methylquinolin-2(1H)-one
- 3-(4-chlorobenzyl)-4-hydroxy-1-methylquinolin-2(1H)-one
- 3,3′-bis(4-chlorobenzylidene)-1,10-methylquinolin-2,20-(1H)-one
Palavra indicadora
Warning
Frases de perigo
Declarações de precaução
Classificações de perigo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órgãos-alvo
Respiratory system
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
dust mask type N95 (US), Eyeshields, Gloves
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
Mechanistic study of electrochemical oxidation of catechols in the presence of 4-hydroxy-1-methyl-2 (1H)-quinolone: Application to the electrochemical synthesis.
Fakhari AR, et al.
Electrochimica Acta, 50(27), 5322-5328 (2005)
Ceric ammonium nitrate (CAN)-mediated oxidative cycloaddition of 1, 3-dicarbonyls to conjugated compounds. Efficient synthesis of dihydrofurans, dihydrofurocoumarins, dihydrofuroquinolinones, dihydrofurophenalenones, and furonaphthoquinone natural products.
Lee YR, et al.
Tetrahedron, 56(45), 8845-8853 (2000)
Iridium catalysed alkylation of 4-hydroxy coumarin, 4-hydroxy-2-quinolones and quinolin-4 (1H)-one with alcohols under solvent free thermal conditions.
Grigg R, et al.
Tetrahedron, 65(36), 7468-7473 (2009)
Michael B Hicks et al.
The Analyst, 142(3), 525-536 (2017-01-18)
The use of a coulometric array detector in tandem with HPLC-UV was evaluated for the absolute quantitation of pharmaceutical compounds without standards, an important capability gap in contemporary pharmaceutical research and development. The high-efficiency LC flow-through electrochemical detector system allows
Mohankumar Saraladevi Resmi et al.
FEBS letters, 589(3), 332-341 (2015-01-06)
Type III polyketide synthases (PKSs) catalyze the biosynthesis of various medicinally important secondary metabolites in plants, but their role in growth and stress response is unclear. Here, we overexpressed quinolone synthase (QNS) from bael in tobacco. QNS-overexpressing plants showed an
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica