PHR1451
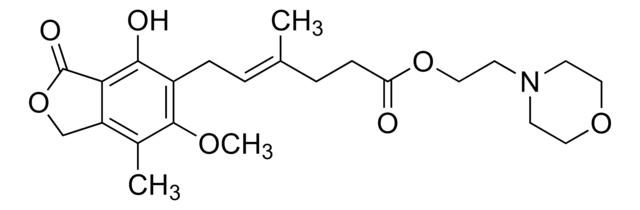
Mycophenolat-Mofetil
Pharmaceutical Secondary Standard; Certified Reference Material
Synonym(e):
Mycophenolat-Mofetil, (4E)-6-(1,3-Dihydro-4-hydroxy-6-methoxy-7-methyl-3-oxo-5-isobenzofuranyl)-4-methyl-4-hexensäure-2-(4-morpholinyl)-ethylester, RS 61443, TM-MMF
About This Item
Empfohlene Produkte
Qualität
certified reference material
pharmaceutical secondary standard
Qualitätsniveau
Agentur
traceable to Ph. Eur. Y0000489
traceable to USP 1448956
API-Familie
mycophenolate
Analysenzertifikat (CofA)
current certificate can be downloaded
Verpackung
pkg of 1 g
Methode(n)
HPLC: suitable
gas chromatography (GC): suitable
Anwendung(en)
pharmaceutical (small molecule)
Format
neat
Lagertemp.
2-30°C
SMILES String
COc1c(C)c2COC(=O)c2c(O)c1C\C=C(/C)CCC(=O)OCCN3CCOCC3
InChI
1S/C23H31NO7/c1-15(5-7-19(25)30-13-10-24-8-11-29-12-9-24)4-6-17-21(26)20-18(14-31-23(20)27)16(2)22(17)28-3/h4,26H,5-14H2,1-3H3/b15-4+
InChIKey
RTGDFNSFWBGLEC-SYZQJQIISA-N
Angaben zum Gen
human ... IMPDH1(3614) , IMPDH2(3615)
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Allgemeine Beschreibung
Mycophenolate mofetil is a prodrug of mycophenolic acid that is utilized as an immunosuppressive agent for the treatment of renal transplant recipients. It may lead to gastrointestinal side effects.
Anwendung
Biochem./physiol. Wirkung
Hinweis zur Analyse
Sonstige Hinweise
Fußnote
Empfohlene Produkte
Ähnliches Produkt
Signalwort
Danger
H-Sätze
Gefahreneinstufungen
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Repr. 1B - STOT RE 1
Lagerklassenschlüssel
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.