Wichtige Dokumente
W288608
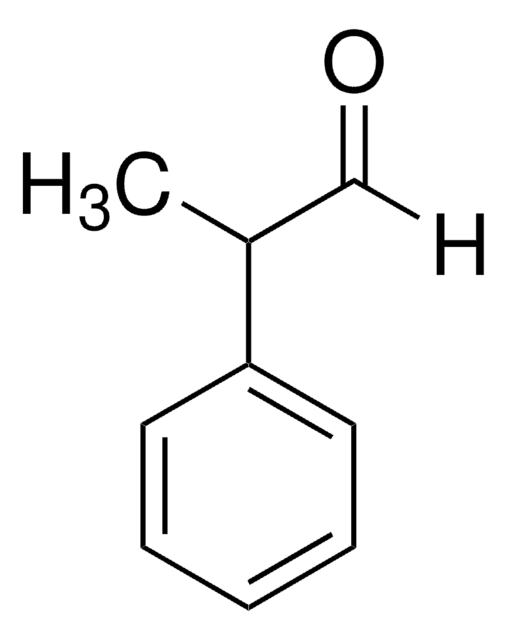
2-Phenylpropionaldehyd
≥95%, FCC, FG
Synonym(e):
2-Phenyl-propanal, Hydratropaldehyd
About This Item
Empfohlene Produkte
Biologische Quelle
synthetic
Qualitätsniveau
Qualität
FG
Halal
Kosher
Agentur
meets purity specifications of JECFA
Einhaltung gesetzlicher Vorschriften
EU Regulation 1334/2008 & 178/2002
FCC
FDA 21 CFR 172.515
Assay
≥95%
Brechungsindex
n20/D 1.517 (lit.)
bp
92-94 °C/12 mmHg (lit.)
Dichte
1.002 g/mL at 25 °C (lit.)
Anwendung(en)
flavors and fragrances
Dokumentation
see Safety & Documentation for available documents
Nahrungsmittelallergen
no known allergens
Organoleptisch
fresh; green; floral
Lagertemp.
2-8°C
SMILES String
[H]C(=O)C(C)c1ccccc1
InChI
1S/C9H10O/c1-8(7-10)9-5-3-2-4-6-9/h2-8H,1H3
InChIKey
IQVAERDLDAZARL-UHFFFAOYSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Allgemeine Beschreibung
Anwendung
- Cytotoxicity, early safety screening, and antimicrobial potential of minor oxime constituents of essential oils and aromatic extracts.: Explores the safety and effectiveness of 2-Phenylpropionaldehyde among other compounds in essential oils, highlighting its potential antimicrobial properties and implications for food safety and preservation (Strub DJ et al., 2022).
- Spectroscopic Evidence for a Cobalt-Bound Peroxyhemiacetal Intermediate.: This study provides spectroscopic evidence of a cobalt-bound intermediate in reactions involving 2-Phenylpropionaldehyde, advancing our knowledge of chemical reaction mechanisms and catalysis (Cho J et al., 2021).
Lagerklassenschlüssel
10 - Combustible liquids
WGK
WGK 1
Flammpunkt (°F)
174.2 °F
Flammpunkt (°C)
79 °C
Persönliche Schutzausrüstung
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.