275875
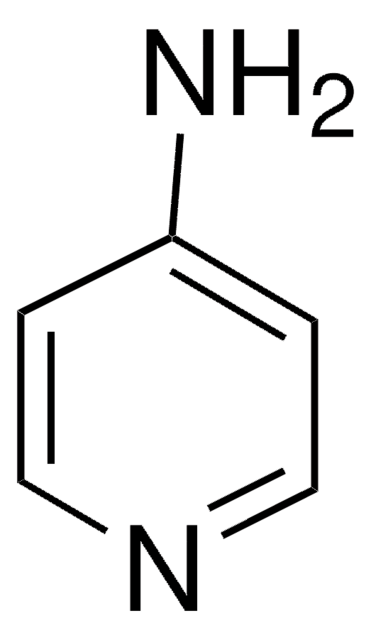
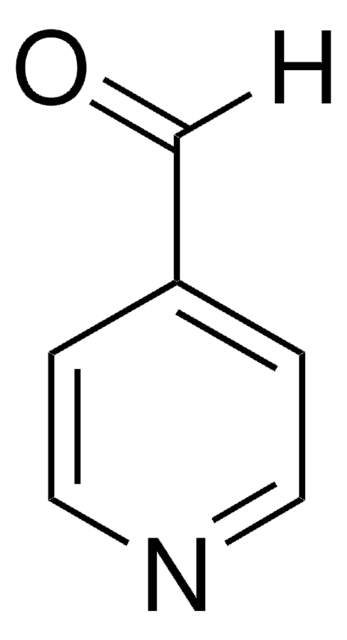
4-Aminopyridine
≥99%
Sinónimos:
4-Pyridinamine, 4-Pyridylamine, 4AP, Fampridine
About This Item
Productos recomendados
Quality Level
assay
≥99%
form
solid
bp
273 °C (lit.)
mp
155-158 °C (lit.)
solubility
DMF: soluble(lit.)
DMSO: soluble(lit.)
THF: soluble(lit.)
acetone: soluble(lit.)
acetonitrile: soluble(lit.)
ethanol: soluble(lit.)
isopropanol: soluble(lit.)
methanol: soluble(lit.)
water: soluble(lit.)
SMILES string
Nc1ccncc1
InChI
1S/C5H6N2/c6-5-1-3-7-4-2-5/h1-4H,(H2,6,7)
InChI key
NUKYPUAOHBNCPY-UHFFFAOYSA-N
Gene Information
human ... KCNA1(3736) , KCNA10(3744) , KCNA2(3737) , KCNA3(3738) , KCNA4(3739) , KCNA5(3741) , KCNA6(3742) , KCNA7(3743) , KCNB1(3745) , KCNB2(9312) , KCNC1(3746) , KCNC2(3747) , KCNC3(3748) , KCNC4(3749) , KCND1(3750) , KCND2(3751) , KCND3(3752) , KCNF1(3754) , KCNG1(3755) , KCNG2(26251) , KCNG3(170850) , KCNG4(93107) , KCNH1(3756) , KCNH2(3757) , KCNH3(23416) , KCNH4(23415) , KCNH5(27133) , KCNH6(81033) , KCNH7(90134) , KCNH8(131096) , KCNQ1(3784) , KCNQ2(3785) , KCNQ3(3786) , KCNQ4(9132) , KCNQ5(56479) , KCNS1(3787) , KCNS2(3788) , KCNS3(3790) , KCNV1(27012) , KCNV2(169522)
mouse ... Kcna3(16491)
rat ... Grin2a(24409)
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
General description
Application
- A reactant to prepare pharmacologically important 4-amino-N-pyridin-4-yl-butyramide hydrochloride.
- A reactant to prepare cinnamic acid derivatives as potent cholinesterase inhibitors.
- A starting material to prepare aza analogs of batracylin, a topoisomerase II inhibitor.
signalword
Danger
Hazard Classifications
Acute Tox. 2 Oral - Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Aquatic Chronic 2 - Eye Dam. 1 - Skin Corr. 1B
Storage Class
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
wgk_germany
WGK 3
flash_point_f
312.8 °F
flash_point_c
156 °C
ppe
dust mask type N95 (US), Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico