594539
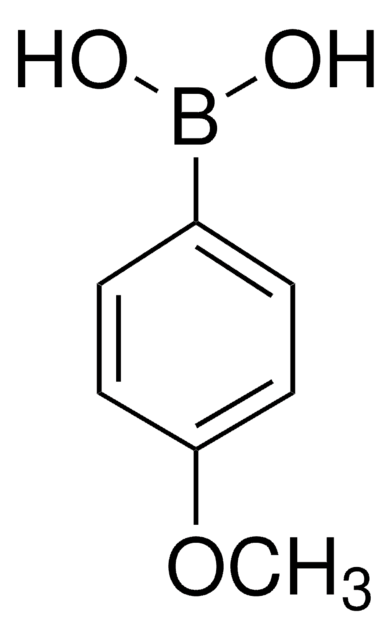
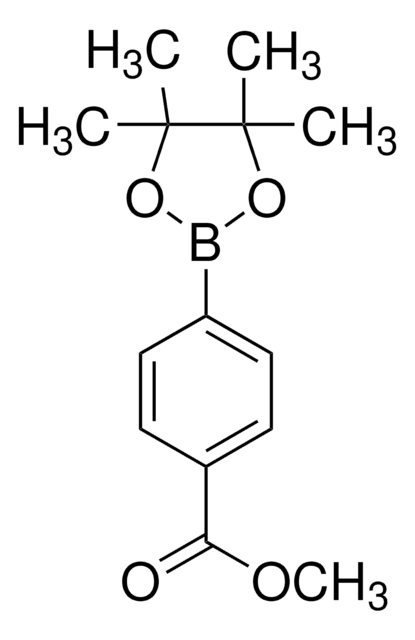
4-Methoxycarbonylphenylboronic acid
≥95%
Sinónimos:
(4-Carbomethoxyphenyl)boronic acid, 4-Carbomethoxybenzeneboronic acid, 4-Methoxycarbonylbenzeneboronic acid, 4-borono-benzoic acid 1-methyl ester, p-(Methoxycarbonyl)boronic acid, p-(Methoxycarbonyl)phenylboronic acid, p-borono-benzoic acid methyl ester, Methyl 4-boronobenzoate, Methyl p-boronobenzoate
About This Item
Productos recomendados
Quality Level
assay
≥95%
form
powder
mp
197-200 °C (lit.)
functional group
ester
SMILES string
COC(=O)c1ccc(cc1)B(O)O
InChI
1S/C8H9BO4/c1-13-8(10)6-2-4-7(5-3-6)9(11)12/h2-5,11-12H,1H3
InChI key
PQCXFUXRTRESBD-UHFFFAOYSA-N
Categorías relacionadas
Application
- Tandem-type Pd(II)-catalyzed oxidative Heck reaction and intramolecular C-H amidation sequence
- Copper-mediated ligandless aerobic fluoroalkylation of arylboronic acids with fluoroalkyl iodides
- One-pot ipso-nitration of arylboronic acids
- Copper-catalyzed nitration
- Cyclocondensation followed by palladium-phosphine-catalyzed Suzuki-Miyaura coupling
- Reagent used in Preparation of
- Biaryls via nickel-catalyzed Suzuki-Miyaura cross-coupling reaction of aryl halides with arylboronic acid†
- Chromenones and their bradykinin B1 antagonistic activit†
- Pt nanoparticles@Photoactive metal-organic frameworks resulting in efficient hydrogen evolution via synergistic photoexcitation and electron injectio†
- Salicylate-based thienylbenzoic acids as E. coli methionine aminopeptidase inhibitor†
Other Notes
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico

![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)