Neural Stem Cell Markers and Antibodies
- Neural Stem Cells
- Neurons, Astrocytes and Oligodendrocyte Cell Types
- NSCs in the Adult Mammalian Brain
- Available NSC, Astrocyte, and Oligodendrocyte Marker Antibodies
Neural Stem Cells
Neural stem cells (NSCs) offer a unique and powerful tool for basic research and regenerative medicine. Changes in the expression levels of specific molecules help to identify the presence of neural stem cells in studies focused on further differentiation toward specific neural lineages. Commonly used markers for neural stem cells include Nestin and SOX2. Other protein markers, expressed at the cell surface are, for example, ABCG2, FGFR1 and Frizzled-9.
Representative Staining of Neurofilament Using Monoclonal Anti-NEFL Antibody (AMAb91314)

Figure 1. The human cerebellum shows strong cytoplasmic immunoreactivity in cell bodies and proximal dendrites of Purkinje cells.

Figure 2. Immunofluorescence staining of mouse striatum shows strong positivity in neural fibers.
Neurons, Astrocytes and Oligodendrocyte Cell Types
The neurons (nerve cells) are electrically excitable cells that transmit and relay the nerve signal within the central nervous system and in the periphery. The neurons communicate via synapses and fall into different functional categories based on their neurochemical properties.

Figure 3. NSCs primarily differentiate into neurons, astrocytes, and oligodendrocytes, depending on environmental cues.
Astrocytes constitute the main glial cell population in the central nervous system. These cells are derived from heterogeneous populations of progenitor cells in the neuroepithelium of the developing central nervous system. Astrocytes play several active roles in the brain, including the secretion or absorption of neural transmitters, ion homeostasis and maintenance of the blood-brain barrier. Oligodendrocytes are glial cells within the CNS and spinal cord whose main function is to form the insulating myelin sheaths around axons in the CNS. Furthermore, oligodendrocytes provide trophic support to neurons by the production of different growth factors like BDNF and GDNF. See tables 1-4 for a list of selected NSC markers.
NSCs in the Adult Mammalian Brain
Neural progenitor stem cells reside in the adult mammalian brain and contribute to brain plasticity throughout life.1 When adult NSCs were initially discovered, it was assumed that their function was to provide a regenerative source for new neurons, and there were hopes that these NSCs might be involved in functional brain repair after injury. Instead, cumulative evidence suggests that the primary function of endogenous adult NSCs is to confer an additional layer of plasticity to the brain via both direct and indirect mechanisms.2 Adult somatic stem cells play a homeostatic role in maintaining tissue organization. Regulation of gene expression by transcription factors represents one fundamental mechanism that controls adult NSCs.3 Though apparently lacking synapses, NSCs express receptors and respond to a variety of neurotransmitters.4 There are two major neurogenic niches in the adult mammalian brain where endogenous NSCs reside, the subventricular zone (SVZ) lining the lateral ventricles and the subgranular zone (SGZ) within the dentate gyrus of the hippocampus. The majority of adult NSCs communicate with other cells via gap junctions and direct cell-cell interactions.5
Representative Staining of Glial Fibrillary Acidic Protein (GFAP) Using the Anti-GFAP Monoclonal Antibody (AMAb91033)

Figure 4. Immunohistochemical staining of the human cerebral cortex shows strong cytoplasmic positivity in astrocytes.

Figure 5. Immunofluorescence staining of rat hippocampus shows strong positivity of the astrocytes.
Representative Staining of Myelin Basic Protein Using the Anti-MBP Monoclonal Antibody (AMAb91063)
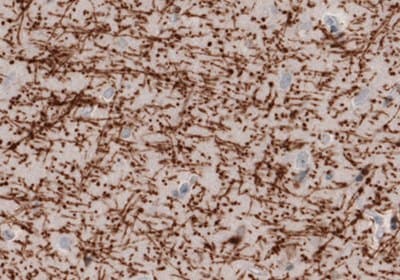
Figure 6. Immunohistochemical staining of the human cerebral cortex shows strong immunoreactivity in myelinated fibers.

Figure 7.Immunofluorescence staining in the rat cerebral cortex shows strong immunoreactivity in myelinated axons.
Neural Stem Cell (NSC) Markers |
|---|
Neuronal Stem Cell Markers |
|---|
Astroglial Markers |
|---|
Neuronal Stem Cell Markers |
|---|
*Products with enhanced validation for the indicated application
For Research Use Only. Not for use in diagnostic procedures.
Unless otherwise stated in the Product(s) specifications, any Antibody product is sold for internal research use only and may not be used for any other purpose, which includes but is not limited to, any commercial, diagnostic, or therapeutic use. Our validation processes pertain only to research uses and do not confirm or assure that our antibodies can be used for any unauthorized uses as set forth herein.
References
To continue reading please sign in or create an account.
Don't Have An Account?