Gene Expression & Silencing
Modulating gene expression is one of the most powerful techniques employed by researchers investigating the complex genetic networks governing biological processes. In particular, gene modulation experiments are used extensively to identify therapeutics effective in combatting diseases ranging from cancer to developmental and neurodegenerative disorders.
Numerous technologies exist for the effective regulation of gene expression in cells and tissues. RNAi (RNA interference) is a natural biological mechanism wherein siRNA (short interfering RNA) duplexes induce potent, targeted inhibition of gene expression. SigmaAldrich® Advanced Genomics has been a leader in developing commercial RNAi products for researchers.
More recently, the development of CRISPR inhibition (CRISPRi) and CRISPR activation (CRISPRa) gives researchers a powerful new set of tools that allows for almost unlimited gene regulation options. Utilizing the unparalleled portfolio of CRISPR products from SigmaAldrich® Advanced Genomics, researchers are able to accurately and efficiently knock-down, knock-out, activate or overexpress virtually any gene.
Featured Categories
Agarose bioreagents cater to cell culture and molecular biology applications, offering ready-to-pour agarose gels for RNA/DNA electrophoresis.

Diagram showing the mechanistic flow of shRNA and siRNA mediated gene silencing in cells.
RNAi - Gene Modulation to Illuminate Genetic Pathways
Functional genomics screens are an integral part of discovery research, allowing you to unravel the mechanisms that belie biological systems, pathways, and diseases. Effective interrogation of these complex molecular interaction networks can be accomplished using shRNA libraries to deliver reliable, efficient, and long-term knockdown.
Sigma-Aldrich® Advanced Genomics is the exclusive provider of the TRC1.5 and TRC2 collections from The RNAi Consortium (TRC). These lentivirus-based shRNA libraries deliver unparalleled coverage and are available in multiple formats that include whole-genome, individual RNAi clones/vectors, and gene family sets.
CRISPRi and CRISPRa - Powerful Gene Inhibition and Activation
CRISPRi (CRISPR interference) and CRISPRa (CRISPR activation) deliver highly efficient silencing and activation of genes, respectively, without altering the underlying DNA sequence. When employed in large scale LOF (loss-of-function) and GOF (gain-of-function) screens, researchers are able to identify unique, yet functionally related, gene pathways that are often missed with other methods.
Visit our document search for data sheets, certificates and technical documentation.

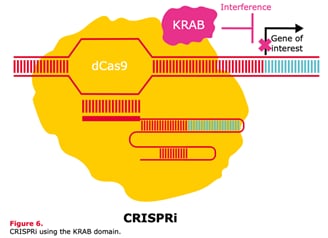
Schematic diagrams of CRISPRa with the Synergystic Activation Mediator complex (SAM) and CRISPRi using the KRAB domain.
Sigma-Aldrich® Advanced Genomics offers a complete suite of optimized CRISPRi and CRISPRa libraries for gene knockdown and overexpression experiments. Pooled CRISPRi and SAM CRISPRa lentiviral libraries are available off-the-shelf or customized to your specific needs.
Wondering if CRISPR is right for your project? Connect with a Sigma-Aldrich® Advanced Genomics Expert.
Related Articles
- We offer advanced design and quality synthesis for predesigned siRNA and siRNA libraries using the Rosetta siRNA Design Algorithm to reduce off-target effects and increase RNAi performance. The algorithm utilizes Position-Specific Scoring Matrices (PSSM) and knowledge of the siRNA seed region to predict the most effective and specific siRNA sequences for a target gene of interest.
- One approach to the analysis of gene expression is to measure the concentration of mRNA of a gene. There are several challenges to such analyses, such as the differences in half life between different transcripts, the temporal patterns of transcription and the lack of correlation between mRNA and protein.
- A series of vectors to enable a successful implementation of your RNAi experiments.
- Enzyme Explorer Product Application Index for Superoxide Dismutase offered by us online. Superoxide Dismutase (SOD) catalyzes the reduction of superoxide anions to hydrogen peroxide.
- Introduction of the modulation in expression profile for some genes and proteins when comparing 2D versus 3D cell culture conditions.
- See All (79)
Related Protocols
- Complete protocol for Lentiviral transduction of suspension cells. This protocol describes the use of MISSION TRC shRNA Lentiviral Particles for long term silencing and phenotypic observation of suspension cells.
- FACS (Fluorescence-Activated Cell Sorting) provides a method for sorting a mixed population of cells into two or more groups, one cell at a time, based on the specific light scattering and fluorescence of each cell. This method provides fast, objective, and quantitative recording of fluorescent signals from individual cells.
- Method for purification, reverse transcription and quantitative PCR for MicroRNAs using Mysticq reagents
- Experimentally determine the appropriate antibiotic concentration for selecting stable cell lines. This titration protocol can determine the lowest concentration of puromycin needed to efficiently select transduced cells.
- Preparation of the Lentiviral Transduction Particles Using Packaging Plasmid Mix
- See All (27)
Find More Articles and Protocols
How Can We Help
In case of any questions, please submit a customer support request
or talk to our customer service team:
Email custserv@sial.com
or call +1 (800) 244-1173
Additional Support
- Chromatogram Search
Use the Chromatogram Search to identify unknown compounds in your sample.
- Calculators & Apps
Web Toolbox - science research tools and resources for analytical chemistry, life science, chemical synthesis and materials science.
- Customer Support Request
Customer support including help with orders, products, accounts, and website technical issues.
- FAQ
Explore our Frequently Asked Questions for answers to commonly asked questions about our products and services.
Related Resources
- /deepweb/assets/sigmaaldrich/marketing/global/documents/145/460/rnai-br7266en-ms.pdf
Depend on our comprehensive shRNA & miRNA portfolio
- Vector Maps
A series of vectors to enable a successful implementation of your RNAi experiments.
- General shRNA FAQs
We answers your frequently asked questions about shRNA, the TRC. MISSION and more.
To continue reading please sign in or create an account.
Don't Have An Account?
