SRP2092
FGF-2 human
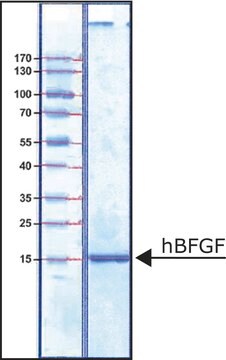
recombinant, expressed in insect cells, ≥85% (SDS-PAGE)
Synonym(s):
BFGF, FGFB, HBGF-2
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
UNSPSC Code:
12352202
NACRES:
NA.26
Recommended Products
biological source
human
recombinant
expressed in insect cells
assay
≥85% (SDS-PAGE)
form
frozen liquid
mol wt
~32.5 kDa
packaging
pkg of 5 μg
storage condition
avoid repeated freeze/thaw cycles
concentration
350 μg/mL
color
colorless to clear
NCBI accession no.
UniProt accession no.
shipped in
dry ice
storage temp.
−70°C
Gene Information
human ... FGF2(2247)
Application
FGF2 (fibroblast growth factor 2)-human has been used to study the role of FGF2 on steroidogenesis and survival of cultured buffalo granulosa cells. It has been used as a control to study activated α2-macroglobulin mediated Müller glial cell migration.
Biochem/physiol Actions
Acidic fibroblast growth factor (FGF-1) and basic fibroblast growth factor (FGF-2) are ubiquitous cytokines found in many tissues. They have effects on multiple cell types derived from mesoderm and neuroectoderm, including endothelial cells. FGF proteins are small peptides of 155 to 268 amino acid residues. The degree of sequence identity between different family members is 30-60 % in a "central domain" of approx. 120 amino acids. This domain confers to FGFs a common tertiary structure and the ability to bind to heparin. Secreted FGFs signal to target cells by binding and activating cell-surface tyrosine kinase FGF receptors (FGFRs; 6, 7). The function of FGFs and FGFRs during embryonic development and adult physiology has been addressed by gain- and loss-of-function experiments in several animal model organisms. These studies have shown that FGFs act as key regulators of developmental events.
Physical form
Clear and colorless frozen liquid solution
Preparation Note
Use a manual defrost freezer and avoid repeated freeze-thaw cycles. While working, please keep sample on ice.
Storage Class
10 - Combustible liquids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Fibroblast growth factor-2.
Nugent MA and Iozzo RV
The International Journal of Biochemistry & Cell Biology, 32, 115-120 (2000)
Li Liu et al.
Biomaterials, 35(24), 6259-6267 (2014-05-09)
Nanofibrous gelatin substrates are suited for long-term expansion of human pluripotent stem cells (hPSCs) under feeder- and serum-free culture conditions. A combinatorial library with different sets of processing parameters was established to assess the culture performance of hPSCs on nanofibrous
L De Moerlooze et al.
Development (Cambridge, England), 127(3), 483-492 (2000-01-13)
The fibroblast growth factor receptor 2 gene is differentially spliced to encode two transmembrane tyrosine kinase receptor proteins that have different ligand-binding specificities and exclusive tissue distributions. We have used Cre-mediated excision to generate mice lacking the IIIb form of
S R Mishra et al.
Research in veterinary science, 108, 98-111 (2016-09-25)
The present study investigated the expression and localization of FGF and its functional receptors in the follicle of buffalo and the treatment of FGF2 on mRNA expression of CYP19A1 (aromatase), PCNA, and BAX (BCL-2 associated X protein) in cultured buffalo
Isako Saga et al.
Neuro-oncology, 16(8), 1048-1056 (2014-05-27)
The metabolic preference of malignant glioma for glycolysis as an energy source is a potential therapeutic target. As a result of the cellular heterogeneity of these tumors, however, the relation between glycolytic preference, tumor formation, and tumor cell clonogenicity has
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service