S4762
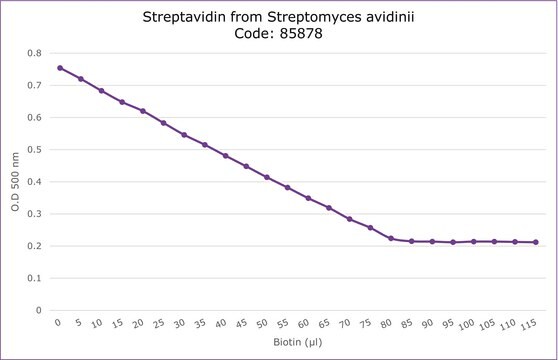
Streptavidin from Streptomyces avidinii
essentially salt-free, lyophilized powder, ≥13 units/mg protein
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Recommended Products
form
essentially salt-free, lyophilized powder
Quality Level
specific activity
≥13 units/mg protein
mol wt
~60 kDa
capacity
4 mol/mol (Biotin)
application(s)
research pathology
storage temp.
−20°C
Looking for similar products? Visit Product Comparison Guide
General description
Streptavidin derives its name from its bacterial source Streptomyces avidinii and from the hen egg-white protein, avidin, which has high affinity to biotin. Its homologous core shares 33% sequence similarity with avidin, as well as sharing a common tetrameric structure. It is a crystalline tetrameric protein, with a molecular weight of 4*15000Da. It binds four molecules of biotin. Streptavidin lacks carbohydrate and sulfur-containing amino acids.
Streptavidin is a crystalline protein isolated from the bacterium Streptomyces avidinii and possesses biotin-binding ability. It has four binding sites for biotin like avidin and binds four molecules of biotin. Streptavidin is now considered as a new type of biotin-binding protein.
Application
Streptavidin from Streptomyces avidinii has been used:
- in wire functionalization
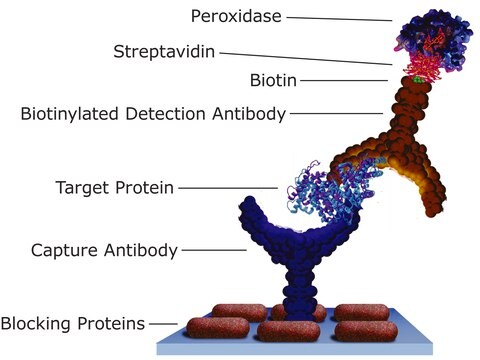
- in streptavidin capture enzyme-linked immunosorbent assay (ELISA)
- in microbubble preparation
Preparation Note
Affinity purified.
Disclaimer
Unless otherwise stated in our catalog or other company documentation accompanying the product(s), our products are intended for research use only and are not to be used for any other purpose, which includes but is not limited to, unauthorized commercial uses, in vitro diagnostic uses, ex vivo or in vivo therapeutic uses or any type of consumption or application to humans or animals.
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Mapping of functional domains in the p22(phox) subunit of flavocytochrome b(559) participating in the assembly of the NADPH oxidase complex by "peptide walking".
Dahan I et al
The Journal of Biological Chemistry, 277(10), 8421-8432 (2002)
A rapid streptavidin-capture ELISA specific for the detection of antibodies to feline foamy virus.
Winkler IG et al
Journal of Immunological Methods, 207(1), 69-77 (1997)
Evaluation of a microwire sensor functionalized to detect Escherichia coli bacterial cells
Lu L and Jun S
Biosensors And Bioelectronics, 36(1), 257-261 (2012)
Biotinylated CdSe/ZnSe nanocrystals for specific fluorescent labeling.
Charvet N et al
Journal of Materials Chemistry, 14.17 , 2638-2642 (2004)
Tilting and wobble of myosin V by high-speed single-molecule polarized fluorescence microscopy.
Beausang JF et al
Biophysical Journal, 104(6), 1263-1273 (2013)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service











![[Nle4,DPhe7]-Biotinyl-α MSH](/deepweb/assets/sigmaaldrich/product/images/175/962/1f49481c-d81e-49b6-83fc-9490335deb42/640/1f49481c-d81e-49b6-83fc-9490335deb42.jpg)
