P4762
Papain from papaya latex
lyophilized powder, ≥10 units/mg protein
Synonym(s):
Papainase
About This Item
Recommended Products
biological source
papaya (latex)
Quality Level
form
lyophilized powder
specific activity
≥10 units/mg protein
mol wt
23.4 kDa
technique(s)
cell culture | mammalian: suitable
single cell analysis: suitable
application(s)
diagnostic assay manufacturing
storage temp.
−20°C
Looking for similar products? Visit Product Comparison Guide
General description
Application
- to digest carotid artery smooth muscle cells
- in dissecting solutions
- to produce Fab fragments of antibodies
- for cell dissociation since it has been shown to be more effective and less damaging with certain tissues
- in the enzyme cocktail for dissociating human fetal retinal tissue
- to digest chondrocytes/scaffold cultures for total DNA quantification for cell proliferation
- as a supplement in neurobasal medium to produce retinal cell suspensions
Biochem/physiol Actions
- The pH optimum is 6.0-7.0
- Papain hydrolyzes esters and amides
pH optimum 6.0-7.0
Also hydrolyzes esters and amides.
Packaging
Unit Definition
Physical form
Preparation Note
Other Notes
inhibitor
substrate
signalword
Danger
hcodes
Hazard Classifications
Eye Irrit. 2 - Resp. Sens. 1 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
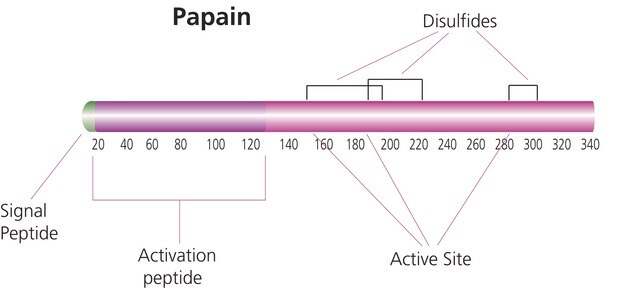
Papain is a cysteine protease of the peptidase C1 family. Papain consists of a single polypeptide chain with three disulfide bridges and a sulfhydryl group necessary for activity of the enzyme.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service