L6785
Lactacystin
≥90% (HPLC), powder, proteosome inhibitor
About This Item
Recommended Products
Product Name
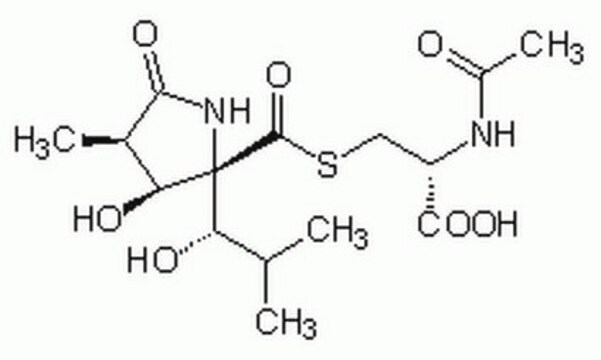
Lactacystin, ≥90% (HPLC)
Quality Level
assay
≥90% (HPLC)
form
powder
potency
4 nM Ki (proteasome inhibitor)
solubility
water: 10 mg/mL, clear, colorless
storage temp.
−20°C
SMILES string
CC(C)[C@H](O)[C@]1(NC(=O)[C@H](C)[C@@H]1O)C(=O)SC[C@H](NC(C)=O)C(O)=O
InChI
1S/C15H24N2O7S/c1-6(2)10(19)15(11(20)7(3)12(21)17-15)14(24)25-5-9(13(22)23)16-8(4)18/h6-7,9-11,19-20H,5H2,1-4H3,(H,16,18)(H,17,21)(H,22,23)/t7-,9+,10+,11+,15-/m1/s1
InChI key
DAQAKHDKYAWHCG-RWTHQLGUSA-N
General description
Application
- as a proteasome inhibitor to inhibit protein degradation
- to inhibit proteasomal activity of cells for live cell imaging
- to block proteasomal proteolysis in human monocyte-derived dendritic cells (MoDCs) for 24 h
- to provide unilateral injection to animals to induce nigrostriatal lesions
Biochem/physiol Actions
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Related Content
DISCOVER Bioactive Small Molecules for Nitric Oxide & Cell Stress Research
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service